Overfitting in Biomedical Method Development: Prevention, Detection, and Correction for Robust Research
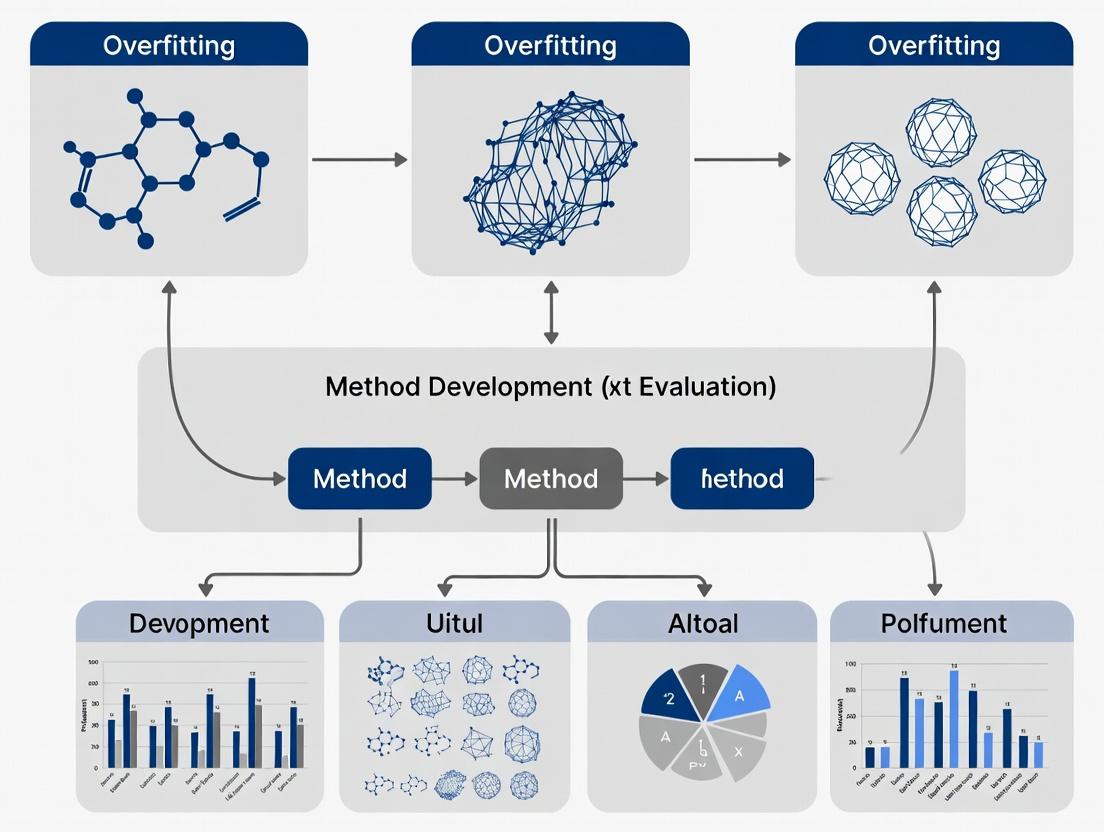
Overfitting poses a critical threat to the validity and reproducibility of biomedical research, especially in modern high-dimensional data settings.
Overfitting in Biomedical Method Development: Prevention, Detection, and Correction for Robust Research
Abstract
Overfitting poses a critical threat to the validity and reproducibility of biomedical research, especially in modern high-dimensional data settings. This article provides a comprehensive guide for researchers and drug development professionals on addressing overfitting throughout the method lifecycle. We first define overfitting and explore its root causes, from p-hacking to data dredging. We then detail robust methodological approaches for prevention, including cross-validation, regularization, and feature selection. The troubleshooting section focuses on detecting overfit models through performance gaps and stability tests. Finally, we cover validation best practices and comparative frameworks to ensure generalizability. The conclusion synthesizes actionable strategies to build more reliable, reproducible, and clinically translatable methods.
What is Overfitting? The Core Concepts and Root Causes in Biomedical Research
Technical Support Center
Troubleshooting Guides & FAQs
Q1: My model achieves >99% accuracy on my training dataset but performs near random (~50%) on the validation set. What is happening and how do I diagnose it? A1: You are experiencing classic overfitting. The model has memorized the training data, including its noise and specific patterns, rather than learning generalizable features.
- Diagnostic Protocol:
- Plot Learning Curves: Generate separate plots for training and validation loss/accuracy vs. training epochs.
- Examine the Gap: A diverging gap where training metric improves while validation metric deteriorates confirms overfitting.
- Conduct Ablation: Systematically remove/reduce model complexity (e.g., layers, units) and observe if the validation performance gap closes.
Q2: In my quantitative structure-activity relationship (QSAR) model for early-stage drug candidates, how can I ensure my validation is meaningful and not just a "lucky" split? A2: Reliable validation in method development requires robust data partitioning and external testing.
- Validation Protocol:
- Stratified Splitting: Use stratified k-fold cross-validation (e.g., k=5 or 10) based on the target variable to maintain distribution.
- Temporal/Scaffold Split: For real-world generalizability, split data by time (older compounds for training) or by molecular scaffold to test predictive power on novel chemotypes.
- Hold-Out External Test Set: Reserve 10-20% of data, completely untouched during model development and hyperparameter tuning, for a final, unbiased performance estimate.
Q3: What concrete regularization techniques are most effective for high-dimensional biological data (e.g., transcriptomics) to prevent overfitting? A3: High-dimensional, low-sample-size data is prone to overfitting. A combination of techniques is required.
- Regularization Protocol:
- L1/L2 Regularization: Apply L1 (Lasso) or L2 (Ridge) penalties to loss functions to shrink coefficients. L1 can drive feature selection to zero.
- Dropout: For neural networks, randomly "drop" a fraction (e.g., 20-50%) of neuron outputs during training to prevent co-adaptation.
- Early Stopping: Monitor validation loss during training and halt iterations when validation loss plateaus or increases for a predetermined number of epochs.
Q4: My deep learning model for microscopy image classification shows excellent validation scores, but fails on new data from a different laboratory. Is this overfitting? A4: This is a form of overfitting to the experimental conditions or data collection bias of your training set, often called "domain shift" or "lack of external validity."
- Mitigation Protocol:
- Data Augmentation: Artificially expand your training set with realistic transformations (rotation, blur, contrast adjustments, simulated staining variations).
- Multi-Source Training: Incorporate data from multiple labs, instruments, and protocols into the training set to force the model to learn invariant features.
- Domain Adaptation: Use techniques like domain adversarial training to explicitly minimize the distributional difference between your source (training) and target (new lab) data.
Table 1: Impact of Regularization Techniques on Model Generalizability (Comparative Study)
| Model Type | Dataset (Sample Size) | Training Accuracy | Validation Accuracy | Test Set Accuracy | Key Regularization Used |
|---|---|---|---|---|---|
| Dense Neural Network | Gene Expression (n=500) | 99.8% | 72.1% | 70.5% | None (Baseline) |
| Dense Neural Network | Gene Expression (n=500) | 95.2% | 88.7% | 87.9% | Dropout (0.5) + L2 |
| Random Forest | Gene Expression (n=500) | 100% | 85.3% | 84.1% | Max Depth Limitation |
| Gradient Boosting | Gene Expression (n=500) | 100% | 89.5% | 88.8% | Early Stopping (Rounds) |
| Convolutional Neural Network | Cell Imaging (n=10,000) | 99.9% | 94.0% | 75.3% | None (Baseline) |
| Convolutional Neural Network | Cell Imaging (n=10,000) | 97.5% | 95.1% | 92.8% | Augmentation + Dropout |
Table 2: Performance Decay Across Data Splits in a QSAR Model
| Data Partition | Number of Compounds | AUC-ROC | Precision | Recall | Description |
|---|---|---|---|---|---|
| Training Set (5-fold CV avg) | 8,000 | 0.95 ± 0.02 | 0.89 | 0.87 | Model development data |
| Internal Validation Set | 1,000 | 0.87 | 0.81 | 0.79 | Held-out from original source |
| Temporal Test Set | 1,000 | 0.82 | 0.75 | 0.78 | Compounds synthesized later |
| External Benchmark Set | 2,500 | 0.76 | 0.69 | 0.72 | Public data from different institution |
Visualizations
Diagram 1: Overfitting Diagnosis via Learning Curves
Diagram 2: Robust Validation Workflow for Method Development
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Robust ML Experimentation in Drug Development
| Item/Reagent | Function & Rationale |
|---|---|
Stratified K-Fold Splitting Module (e.g., scikit-learn StratifiedKFold) |
Ensures representative class distribution in each fold, preventing bias in cross-validation estimates. |
| L1/L2 Regularization Optimizers (e.g., AdamW, SGD with weight decay) | Optimizers with built-in weight decay explicitly penalize complex models, promoting simpler, more generalizable solutions. |
| Data Augmentation Pipeline (e.g., Albumentations, Torchvision transforms) | Simulates experimental variance (noise, rotation, scaling) to artificially expand training data and improve invariance. |
Early Stopping Callback (e.g., Keras EarlyStopping, PyTorch Lightning EarlyStopping) |
Monitors validation metric and automatically halts training when overfitting begins, preventing waste of compute resources. |
Molecular Scaffold Split Library (e.g., RDKit Bemis-Murcko scaffold generation) |
Enables splitting datasets by core molecular structure to rigorously test predictive power on novel chemotypes. |
| External Benchmark Datasets (e.g., ChEMBL, PubChemQC, MoleculeNet) | Provides completely independent, publicly available data for the final, critical test of a model's generalizability. |
| Domain Adaptation Framework (e.g., DANN - Domain-Adversarial Neural Networks) | Explicitly reduces distribution shift between source (training) and target (new lab/assay) data domains. |
Technical Support Center: Troubleshooting Guide & FAQs for Method Validation in Biomedical Research
Context: This support center addresses common pitfalls in method development and evaluation, framed within the critical thesis of addending overfitting to ensure robust, translatable research outcomes.
FAQs: Addressing Overfitting and Validation
Q1: Our clinical prediction model has excellent AUC (>0.95) on our training cohort but fails completely on a validation set from a different clinic. What are the most likely causes and steps to diagnose them?
A: This is a classic sign of overfitting and dataset shift. Follow this diagnostic protocol:
- Perform Feature Importance Analysis: Compare the top 10 features by weight/coefficient in your model between the training and validation set performances. Overfit models often rely on cohort-specific technical artifacts (e.g., batch-specific biomarkers).
- Conduct Data Drift Analysis:
- Protocol: For each feature (especially top model features), use a two-sample Kolmogorov-Smirnov test to compare distributions between training and validation sets. Create a table of p-values and effect sizes (Cohen's d).
- Interpretation: Significant drift (p < 0.01, d > 0.5) in key features indicates non-generalizable data.
- Simplify the Model: Retrain a model with drastically reduced complexity (e.g., reduce polynomial terms, increase regularization strength) on the training set and re-test on the validation set. If performance stabilizes, overfitting is confirmed.
Q2: During biomarker identification from high-dimensional proteomics data, we get hundreds of significant candidates. How do we triage these to find the few that are biologically plausible and not statistical artifacts?
A: This requires a multi-stage filtering protocol to append robustness to the discovery.
- Apply Strict Multiple Testing Correction: Use the Benjamini-Hochberg procedure to control the False Discovery Rate (FDR). Start with an FDR < 0.01.
- Require Biological Replication: Insist that candidates must be significant in at least two independent cohorts or experimental batches.
- Integrate External Knowledge:
- Protocol: Use enrichment analysis (e.g., via Enrichr API) against pathways (KEGG, Reactome) and disease ontologies. Also, query protein-protein interaction databases (STRING).
- Action: Prioritize biomarkers that cluster in known disease-relevant pathways or have documented interactions.
Q3: Our drug screening assay shows high Z' factor (>0.7) in validation but yields inconsistent results when used for novel compound testing. What should we check?
A: High Z' confirms assay robustness but not its biological relevance or susceptibility to interference.
- Check for Assay Interference: For novel compounds, run counter-screens.
- Protocol: For luminescence-based assays, run a luciferase interference assay. For fluorescence-based, test for autofluorescence or quenching. Include a control well with compound and reporter enzyme only (no cells).
- Verify Target Engagement: An assay may be precise but not measuring the intended biology.
- Protocol: Use a cellular thermal shift assay (CETSA) or drug affinity responsive target stability (DARTS) to confirm that your lead compounds physically bind the intended target protein in a cellular context.
- Assess Contextual Specificity: Test the assay in a genetically modified cell line where the target gene is knocked out. A true signal should be abolished.
Table 1: Impact of Overfitting Mitigation Strategies on Model Performance
| Strategy | Training AUC (Mean ± SD) | Hold-Out Test AUC (Mean ± SD) | Generalization Improvement (ΔAUC) |
|---|---|---|---|
| No Regularization (Base) | 0.98 ± 0.02 | 0.65 ± 0.10 | 0.00 (Reference) |
| L2 Regularization Added | 0.92 ± 0.03 | 0.78 ± 0.06 | +0.13 |
| Feature Selection + Regularization | 0.88 ± 0.04 | 0.82 ± 0.05 | +0.17 |
| External Validation Cohort | 0.87 ± 0.05 | 0.81 ± 0.05 | +0.16 |
Table 2: Biomarker Verification Success Rates by Stage
| Validation Stage | Number of Candidates Input | Candidates Confirmed | Success Rate | Typical Cost & Time |
|---|---|---|---|---|
| Discovery (Omics) | 50,000+ | 200-500 | ~1% | High, 3-6 months |
| Analytical Verification (ELISA/MS) | 200 | 50 | 25% | Medium, 2-4 months |
| Clinical Validation (2+ Cohorts) | 50 | 2-5 | 4-10% | Very High, 1-3 years |
Experimental Protocols
Protocol 1: Nested Cross-Validation for Robust Clinical Model Development
- Objective: To provide an unbiased estimate of model performance while optimizing hyperparameters, effectively addending overfitting.
- Materials: Dataset with labels, machine learning environment (e.g., Python/scikit-learn, R/caret).
- Method:
- Define an outer loop (e.g., 5-fold CV) for performance estimation.
- Within each outer training fold, define an inner loop (e.g., 3-fold CV) for hyperparameter optimization (e.g., grid search over regularization strength, tree depth).
- Train a model with the optimal inner-loop parameters on the entire outer training fold.
- Test this model on the held-out outer test fold. Record performance metric (AUC, accuracy).
- Repeat for all outer folds. The mean performance across all outer test folds is the final, robust estimate.
Protocol 2: Orthogonal Validation of a Putative Biomarker via SRM/MRM Mass Spectrometry
- Objective: To transition a biomarker candidate from a discovery platform (e.g., shotgun proteomics) to a validated, quantitative assay.
- Materials: Patient samples (discovery and independent cohort), synthetic stable isotope-labeled peptide standard for the biomarker, triple-quadrupole LC-MS/MS system.
- Method:
- Peptide Selection: From discovery data, select 2-3 proteotypic peptides unique to the target protein.
- Assay Development: Optimize MS parameters (collision energy, precursor/product m/z transitions) for each peptide and its labeled counterpart.
- Quantification: Spike a known amount of labeled standard into each digested patient sample. Run SRM/MRM assay.
- Analysis: Calculate the ratio of endogenous peptide peak area to labeled standard peak area. Use a calibration curve (if available) for absolute quantification, or report relative ratios across samples.
Visualizations
Diagram 1: The Impact of Overfitting on Research Translation
Diagram 2: Robust Biomarker Identification Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
| Item/Category | Example Product/Source | Primary Function in Mitigating Overfitting |
|---|---|---|
| Stable Isotope-Labeled Standards | SIS peptides (Sigma, JPT), AQUA peptides | Provides internal controls for MS-based biomarker verification, enabling accurate quantification and reducing technical variance. |
| Validated Antibody Panels | Luminex Assay Kits, R&D Systems DuoSet ELISA | Ensures specificity in immunoassays for biomarker validation, critical for reproducible results across labs. |
| Reference Cell Lines & Controls | ATCC CRISPR-modified isogenic lines, Coriell Institute biobank | Provides genetically defined controls for drug screening assays to confirm on-target activity and reduce false positives. |
| Chemical Probes (with controls) | Selleckchem probe sets, Tocris Biotools tool compounds | High-quality, selective small molecules used to validate drug targets; paired inactive analogs control for off-target effects. |
| High-Quality Biobanked Samples | Independent, well-annotated clinical cohorts (e.g., UK Biobank, TCGA) | Essential for external validation of models and biomarkers, providing the ultimate test against overfitting to a single dataset. |
| Benchmarking Datasets | MLRepo, PMLB, CAMDA challenges | Pre-curated public datasets for testing and comparing algorithm performance in a standardized, unbiased manner. |
Technical Support Center
Troubleshooting Guides & FAQs
Q1: My initial analysis yielded a null result, but after testing multiple alternative model specifications, I found one with p < 0.05. Is this a valid finding? A: This is a classic symptom of p-hacking (also known as selective inference). The reported p-value is invalid because it does not account for the multiple comparisons (model tests) performed. The probability of finding at least one statistically significant result by chance increases with each test you run.
- Troubleshooting Protocol: Use pre-registration. Before collecting or looking at your data, document your primary hypothesis, analysis plan, and model specification in a public registry. Adhere strictly to this plan for your primary analysis. For exploratory analyses, use correction methods (e.g., Bonferroni, Benjamini-Hochberg) and clearly label them as such.
Q2: I have a large dataset with hundreds of variables. How can I efficiently find significant correlations for my drug response outcome? A: Blindly testing all possible associations is data dredging (or "fishing"). It will almost certainly produce false positive associations due to chance alone, especially in high-dimensional data.
- Troubleshooting Protocol:
- Split Your Data: Before any exploration, randomly split your data into a discovery set (e.g., 70%) and a validation set (e.g., 30%). Lock the validation set away.
- Explore on Discovery Set: Conduct your exploratory analysis (e.g., correlation scans, machine learning feature selection) only on the discovery set.
- Confirm on Validation Set: Test only the hypotheses or models generated in step 2 on the pristine validation set. The validation-set p-value is a more honest estimate of true performance.
Q3: My gene expression biomarker panel shows perfect classification in my training set (n=20 samples, p=500 genes), but fails completely in an independent test. What went wrong? A: You have encountered the "High-Dimensional, Low-Sample-Size" (HDLSS) curse. With far more features (p) than samples (n), models can easily find spurious patterns that fit the noise in your specific small sample, leading to catastrophic overfitting and failure to generalize.
- Troubleshooting Protocol: Apply dimensionality reduction and regularization.
- Feature Selection: Use independent biological knowledge or univariate screening with extremely stringent criteria to reduce the feature set before modeling.
- Regularized Modeling: Employ algorithms designed for HDLSS contexts (e.g., Lasso regression, Ridge regression, Elastic Net). These techniques penalize model complexity to prevent overfitting.
- Use Cross-Validation Correctly: Perform all feature selection and model tuning steps within each fold of cross-validation on the training data only to avoid leakage.
Q4: How do I choose the right multiple testing correction for my high-throughput screen? A: The choice depends on your goal: controlling the Family-Wise Error Rate (FWER) is stricter, while controlling the False Discovery Rate (FDR) is more common in exploratory omics studies.
Table: Multiple Testing Correction Methods
| Method | Controls For | Best Use Case | Key Consideration |
|---|---|---|---|
| Bonferroni | Family-Wise Error Rate (FWER) | Confirmatory studies with a small number of pre-planned tests. Very conservative. | Over-corrects, leading to many false negatives. Adjusted threshold = α / m (m=# of tests). |
| Benjamini-Hochberg | False Discovery Rate (FDR) | Exploratory high-dimensional studies (genomics, proteomics). Less conservative. | Controls the proportion of significant results that are false positives. More powerful than Bonferroni. |
| Permutation-Based FDR | False Discovery Rate (FDR) | Complex dependency structures between tests (e.g., GWAS, imaging). | Computationally intensive but makes fewer assumptions about test distribution and independence. |
Q5: What are essential experimental design reagents and tools to mitigate overfitting from the start? A: The Scientist's Toolkit for Robust Research
| Research Reagent Solution | Function in Mitigating Overfitting |
|---|---|
| Pre-Registration Template | Forces explicit a priori specification of hypotheses, primary outcomes, and analysis plans, neutralizing p-hacking. |
| Independent Validation Cohort | Provides an unbiased estimate of model performance and generalizability. The gold standard for method evaluation. |
| Data/Code Repository Access | Enables full transparency, peer auditability, and reproducibility of all analysis steps, reducing hidden flexibility. |
| Power/Sample Size Calculator | Ensures studies are designed with adequate sample size to detect effects, reducing the temptation to dredge underpowered data. |
| Regularized ML Software (e.g., glmnet) | Provides built-in algorithms (Lasso, Ridge) that prevent overfitting in high-dimensional data during model development. |
| High-Performance Computing (HPC) Access | Enables the use of computationally intensive but honest validation methods like permutation testing and nested cross-validation. |
Experimental Protocols
Protocol 1: Nested Cross-Validation for HDLSS Model Development Purpose: To provide an unbiased performance estimate for a model that requires both feature selection and hyperparameter tuning.
- Outer Loop (Performance Estimation): Split data into k folds (e.g., k=5 or 10).
- Hold Out One Outer Fold: This fold serves as the temporary test set.
- Inner Loop (Model Selection): On the remaining (k-1) folds, perform a second, independent cross-validation.
- Feature Selection & Tuning: Within the inner loop, perform all steps (e.g., feature filtering, selecting the LASSO lambda) repeatedly. Find the optimal model configuration.
- Train Final Inner Model: Train a new model using this optimal configuration on all (k-1) outer-loop training folds.
- Test: Apply this final model to the held-out outer test fold (from step 2) to get one unbiased performance score.
- Repeat: Iterate so each outer fold serves as the test set once. The average performance across all outer folds is the final validated estimate.
Protocol 2: Permutation Test for Assessing Significance Without Overfitting Purpose: To generate a valid null distribution for any complex test statistic (e.g., classifier AUC, biomarker correlation) in the context of small samples or complex data.
- Calculate Real Statistic: Compute your test statistic (e.g., AUC) on your real dataset with true labels.
- Permute Labels: Randomly shuffle the outcome labels (or treatment assignments) of your dataset, breaking the true relationship between variables and outcome.
- Calculate Null Statistic: Re-compute the same test statistic on this permuted, null dataset.
- Repeat: Perform steps 2-3 a large number of times (e.g., 10,000) to build a robust null distribution of the test statistic under the assumption of no effect.
- Calculate p-value: The empirical p-value = (number of permutations where the null statistic ≥ real statistic + 1) / (total permutations + 1). This gives a valid, non-parametric p-value.
Visualizations
Title: Data Splitting Workflow to Prevent Overfitting
Title: Consequences of the HDLSS Curse
Technical Support Center: Overfitting Prevention & Diagnostic Tools
Frequently Asked Questions (FAQs)
Q1: My model performs excellently on my training dataset but fails on new, external validation data. What is the most likely cause and how do I diagnose it? A: This is the hallmark symptom of overfitting. The model has learned noise or specific patterns unique to your training set rather than generalizable biological relationships.
- Diagnostic Steps:
- Split Your Data: Before any analysis, randomly split your data into three sets: Training (60-70%), Validation (15-20%), and Hold-out Test (15-20%). Use the hold-out test set only once at the very end.
- Learning Curves: Plot model performance (e.g., R², AUC) for both training and validation sets against increasing model complexity or training iterations. A growing gap between curves indicates overfitting.
- Apply Regularization: Implement techniques like Lasso (L1) or Ridge (L2) regression that penalize overly complex models.
Q2: In high-throughput 'omics studies (genomics, proteomics), I have thousands of features (p) but only tens of samples (n). How can I avoid false discoveries? A: The "p >> n" problem is a primary driver of overfitting in modern biology.
- Diagnostic & Prevention Protocol:
- Apply Dimensionality Reduction: Use Principal Component Analysis (PCA) or use domain knowledge for feature selection before predictive modeling.
- Use Cross-Validation Correctly: Employ nested cross-validation, where an inner loop performs feature selection and hyperparameter tuning, and an outer loop provides an unbiased performance estimate. Never use the same cross-validation loop for both feature selection and final performance estimation.
- Adjust for Multiple Testing: Apply Benjamini-Hochberg (False Discovery Rate) or Bonferroni corrections to p-values from many simultaneous hypotheses.
Q3: How can I tell if my 'statistically significant' biomarker is a result of overfitting to cohort-specific noise? A: Implement rigorous external validation.
- Experimental Validation Protocol:
- Technical Replication: Re-measure the biomarker in the same samples using a different technical platform (e.g., different antibody lot, different mass spectrometry run).
- Biological Replication: Test the biomarker in an independent cohort of patients/samples, collected and processed by a different team if possible.
- Experimental Perturbation: If possible, use in vitro or in vivo models to functionally perturb the identified target and see if the predicted biological effect holds.
Q4: What are the best practices for reporting methods to ensure my work is reproducible and not overfit? A: Transparency is key. Adhere to community reporting standards (e.g., MIAME, ARRIVE, STROBE).
- Checklist for Submission:
- Clearly state how and when the data was split into training/validation/test sets.
- Provide all code and software version numbers in a public repository (e.g., GitHub, Zenodo).
- Report all hyperparameters tried during tuning, not just the final selected ones.
- For preclinical studies, publish negative results and studies that failed to replicate initial findings.
Troubleshooting Guide: Common Overfitting Scenarios
| Scenario | Symptoms | Root Cause | Corrective Action |
|---|---|---|---|
| Biomarker Discovery | A 20-gene signature has 95% accuracy in the discovery cohort but <60% in a similar published cohort. | Feature selection was performed on the entire dataset without a hold-out set. | Re-analyze using a completely independent validation cohort or simulate one via rigorous nested CV. |
| Dose-Response Modeling | A complex polynomial model fits the training dose-response data perfectly but produces nonsensical predictions for interpolated doses. | Model complexity (degree of polynomial) is too high for the number of data points. | Use a simpler model (e.g., 4-parameter logistic curve) or apply Bayesian regularization to constrain parameters. |
| High-Content Imaging Analysis | A deep learning model accurately classifies treatment effects in images from one lab but fails on images from another using a different microscope. | The model overfit to lab-specific image artifacts (background, staining intensity). | Use data augmentation (rotations, flips, noise injection) and incorporate images from multiple sources in the training set. |
Quantitative Data Summary: Impact of Overfitting Mitigation Strategies
Table 1: Effect of Validation Strategy on Reported Model Performance in Published Studies (Simulated Meta-Analysis Data)
| Validation Strategy | Average Reported AUC | Average Performance Drop in External Validation | Estimated Risk of Non-Reproducibility |
|---|---|---|---|
| None (Resubstitution) | 0.95 | -0.25 | Very High |
| Simple Train/Test Split (80/20) | 0.87 | -0.15 | High |
| 10-Fold Cross-Validation | 0.85 | -0.12 | Moderate |
| Nested Cross-Validation | 0.83 | -0.05 | Low |
| Independent External Cohort | 0.80 | N/A (This is the validation) | Very Low |
Table 2: Impact of Multiple Testing Correction on Significant Hits in a Genomic Study (Example: 10,000 genes tested)
| Analysis Method | Nominal p-value threshold | Uncorrected Significant Hits | FDR-Adjusted (q<0.05) Significant Hits | Approx. False Positives |
|---|---|---|---|---|
| No Correction | p < 0.05 | ~500 | N/A | ~500 |
| Bonferroni Correction | p < 5e-6 | 15 | 12 | ~0.05 |
| Benjamini-Hochberg (FDR) | q < 0.05 | 110 | 110 | ~5 |
Experimental Protocol: Nested Cross-Validation for Predictive Modeling
Objective: To obtain an unbiased estimate of a predictive model's performance while optimizing hyperparameters and/or selecting features.
Materials: Dataset with features (X) and outcome labels (y). Computational environment (e.g., Python/R).
Procedure:
- Define Outer Loop (Performance Estimation): Split the entire dataset into k outer folds (e.g., k=5 or 10).
- Iterate Outer Loop: For each outer fold i: a. Set aside fold i as the outer test set. b. The remaining k-1 folds constitute the outer training set.
- Define Inner Loop (Model Tuning): On the outer training set, perform a second, independent k-fold cross-validation (the inner loop).
- Iterate Inner Loop: For each combination of hyperparameters/feature sets: a. Train the model on the inner training folds. b. Evaluate it on the inner validation fold. c. Average performance across all inner folds to select the best hyperparameter/feature set.
- Train Final Inner Model: Train a new model on the entire outer training set using the optimal hyperparameters identified in Step 4.
- Evaluate on Outer Test Set: Apply this final model to the held-out outer test set (fold i) to obtain an unbiased performance score.
- Aggregate Results: Repeat steps 2-6 for all k outer folds. The average performance across all outer test sets is the final, robust performance estimate.
Visualization: Workflows and Relationships
Title: Robust Model Development & Evaluation Workflow
Title: How Overfitting Contributes to the Reproducibility Crisis
The Scientist's Toolkit: Research Reagent Solutions for Validation
| Reagent/Tool Category | Specific Example | Primary Function in Combatting Overfitting |
|---|---|---|
| Reference Standards | Certified cell lines (e.g., from ATCC), synthetic peptide standards, control plasmids. | Provides a consistent baseline across experiments and labs, enabling technical replication and detection of batch effects. |
| Validated Assay Kits | FDA-approved IVD kits, PMA-approved companion diagnostics. | Uses rigorously optimized and locked-down protocols to minimize technical variability that can be mistaken for signal. |
| Knockout/Knockdown Tools | CRISPR-Cas9 kits, validated siRNA pools, isogenic cell line pairs. | Enforces causal validation of putative biomarkers or targets identified in correlative models, moving beyond prediction. |
| Chemical Probes | High-quality, selective kinase inhibitors; well-characterized agonists/antagonists. | Allows pharmacological perturbation to test predictions from computational models in biological systems. |
| Data & Code Repositories | GEO, PRIDE, GitHub, Zenodo, Synapse. | Facilitates independent re-analysis and validation of published models, exposing overfit patterns. |
Troubleshooting Guides & FAQs
General Model Development Issues
Q1: My model performs excellently on training data but fails on new test sets. What is the root cause and how can I fix it? A: This is a classic symptom of overfitting, where a model has high variance and low bias. The model has learned the noise and specific patterns in the training data rather than the generalizable signal.
- Troubleshooting Steps:
- Diagnose: Plot learning curves (train vs. validation error vs. model complexity/epochs).
- Regularize: Apply L1 (Lasso) or L2 (Ridge) regularization to penalize overly complex coefficients.
- Simplify: Reduce model complexity (e.g., decrease polynomial degree, reduce nodes/layers in a neural network).
- Augment Data: Use data augmentation techniques or collect more diverse training data.
- Use Cross-Validation: Implement k-fold cross-validation to get a more robust estimate of performance.
Q2: How do I know if my model is too simple (underfitting) or appropriately complex? A: Underfit models exhibit high bias and low variance; they perform poorly on both training and validation data.
- Troubleshooting Steps:
- Diagnose: If training error is unacceptably high, the model is likely underfitting.
- Increase Complexity: Add relevant features, increase polynomial degree, or add capacity to your algorithm.
- Reduce Regularization: Decrease the strength of regularization parameters.
- Train Longer: For iterative models like neural networks, increase the number of training epochs.
Q3: What is the "double-dipping" problem, and how can I avoid it in my analysis? A: Double-dipping (or circular analysis) occurs when the same data is used for both hypothesis generation (e.g., feature selection) and hypothesis testing (e.g., model evaluation), leading to optimistically biased results and inflated false-positive rates.
- Troubleshooting Protocol:
- Use a Hold-Out Set: Before any analysis, split data into three sets: Training, Validation (for tuning), and a completely held-out Test set used only once for final evaluation.
- Employ Nested Cross-Validation: Use an outer loop for performance estimation and an inner loop for model/feature selection. This keeps the selection process separate from the final evaluation.
- Pre-register Analysis Plans: Define your model, features, and evaluation metrics before observing the test data.
Table 1: Impact of Model Complexity on Error Components
| Model Complexity Level | Typical Training Error | Typical Validation Error | Dominant Error Type | Indicated Problem |
|---|---|---|---|---|
| Very Low | High | Very High | Bias | Severe Underfitting |
| Low | Medium-High | High | Bias | Underfitting |
| Optimal | Low | Low (minimized) | Balanced | Well-Fitted Model |
| High | Very Low | Medium-High | Variance | Overfitting |
| Very High | Extremely Low | Very High | Variance | Severe Overfitting |
Table 2: Common Remedies for Model Fitting Problems
| Problem | Primary Cause | Recommended Solutions (in order of priority) |
|---|---|---|
| Overfitting | High Variance | 1. Gather more training data.2. Apply regularization (L1/L2/Dropout).3. Reduce model complexity.4. Use ensemble methods (e.g., bagging). |
| Underfitting | High Bias | 1. Increase model complexity (features, parameters).2. Train for more iterations/epochs.3. Reduce regularization strength.4. Use a more advanced model algorithm. |
| Double-Dipping Bias | Data Leakage | 1. Implement strict train/validation/test splits.2. Use nested cross-validation.3. Perform independent validation on a fresh cohort. |
Experimental Protocols
Protocol 1: Nested Cross-Validation to Prevent Double-Dipping Objective: To obtain an unbiased estimate of model performance when both hyperparameter tuning and feature selection are required.
- Data Partitioning: Start with your full dataset.
- Outer Loop (Performance Estimation): Split data into k outer folds. For each outer fold: a. Designate the fold as the temporary test set. b. The remaining k-1 folds form the development set.
- Inner Loop (Model Selection): On the development set, perform a second, independent m-fold cross-validation. a. Use this inner loop to train and tune different model configurations (e.g., varying hyperparameters, feature subsets). b. Select the best-performing configuration based on the inner validation scores.
- Final Evaluation: Train the selected configuration on the entire development set. Evaluate it on the held-out temporary test set from step 2a.
- Aggregation: Repeat for all k outer folds. The average score across all temporary test sets provides the final, unbiased performance estimate.
Protocol 2: Learning Curve Analysis for Bias-Variance Diagnosis Objective: To visually diagnose whether a model suffers from high bias or high variance and guide resource allocation.
- Setup: Define a model architecture and evaluation metric (e.g., RMSE, Accuracy).
- Iterative Training: For n different training set sizes (e.g., 10%, 20%, ..., 100% of available data): a. Randomly sample a subset of the training data of the specified size. b. Train the model on this subset. c. Calculate the error/score on: i) the training subset, and ii) a fixed, held-out validation set.
- Plotting: Plot both the training error and validation error as a function of training set size.
- Interpretation:
- Converging High Errors: Both curves plateau at a high error → High Bias.
- Large Gap: Training error is low, validation error is significantly higher and the gap doesn't close with more data → High Variance.
- Converging Low Errors: Both curves converge to a low error → Ideal.
Visualizations
Bias-Variance Tradeoff Relationship
Nested Cross-Validation Workflow to Avoid Double-Dipping
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Robust Method Development & Evaluation
| Item | Category | Function in Addressing Overfitting & Bias |
|---|---|---|
| Scikit-learn | Software Library | Provides built-in functions for train/test splits, cross-validation (including nested), and regularization, enforcing proper workflow. |
| MLflow / Weights & Biases | Experiment Tracking | Logs all hyperparameters, data splits, and metrics for every run, ensuring reproducibility and audit trails to detect data leakage. |
| Matplotlib / Seaborn | Visualization | Creates essential diagnostic plots (learning curves, validation curves, feature importance) to visualize bias-variance. |
| DVC (Data Version Control) | Data Management | Versions datasets and model artifacts, guaranteeing the exact data split used for a model can be recovered and validated. |
| Pre-registration Template | Documentation | A structured document to define hypotheses, analysis plans, and model specifications before data analysis begins, mitigating double-dipping. |
| Statistical Test Suites | Analysis Toolkits | Libraries (e.g., statsmodels, scipy) for calculating p-values and confidence intervals on held-out test sets only, preventing inflation. |
| Public Benchmark Datasets | Reference Data | Well-curated datasets (e.g., from TCGA, PubChem) with standard splits allow for fair comparison and baseline establishment. |
Building Defenses: Proactive Methodological Strategies to Prevent Overfitting
Technical Support Center
Troubleshooting Guides & FAQs
Q1: My model performs excellently on the validation set but fails on the test set. What is the most likely cause? A: This is a classic sign of data leakage or an improper splitting protocol. The validation set is likely not representative or has been used to influence training decisions repeatedly, causing overfitting to the validation set. Ensure your initial data split (Train/Val/Test) is performed before any preprocessing or feature selection, using a method that preserves the distribution of your target variable (e.g., stratified splitting for classification). The test set must be locked away and used for a single, final evaluation.
Q2: How do I partition my dataset when I have temporal or batch-specific effects (e.g., multi-center clinical trial data)? A: For data with inherent grouping or temporal structure, a simple random split violates the independence assumption. Use group-based or time-based splitting.
- Protocol: Identify the grouping variable (e.g., patient ID, clinical trial site, experimental batch). Use
GroupShuffleSplitorGroupKFold(from scikit-learn) to ensure all samples from the same group are contained within a single split (train, validation, or test). This prevents the model from learning group-specific artifacts and gives a true estimate of performance on new groups.
Q3: What is the minimum recommended size for a test set to be statistically meaningful? A: There is no universal rule, but guidelines exist based on desired precision. A common heuristic is to allocate 10-20% of your total data to the test set, provided this yields a sufficient absolute sample size. For performance metrics like accuracy or AUC, the required size depends on the expected variance.
Table 1: Minimum Test Set Sizes for Desired Confidence Interval Width (Binary Classification, ~80% Accuracy)
| Confidence Level | Target CI Width (±) | Minimum Test Set Size (n) |
|---|---|---|
| 95% | 0.05 | ~246 |
| 95% | 0.03 | ~683 |
| 95% | 0.02 | ~1537 |
Protocol for Sizing: Use power analysis for proportions. Formula for accuracy: n = (Z^2 * p * (1-p)) / d^2, where Z is the Z-score (e.g., 1.96 for 95% CI), p is the expected accuracy, and d is the margin of error (CI half-width).
Q4: How should I handle class imbalance when creating splits for a rare event prediction task? A: Use stratified splitting. This maintains the relative proportion of each class across the train, validation, and test sets. This is crucial to prevent a scenario where a rare class is underrepresented or absent in a split, skewing performance estimates.
- Protocol: Utilize
StratifiedShuffleSplitorStratifiedKFold. Provide the target variable (y) to the splitting function. Ensure that the minority class has enough representatives in the validation and test sets to compute meaningful metrics (e.g., precision, recall).
Q5: What is nested cross-validation, and when is it mandatory? A: Nested cross-validation (CV) is the gold-standard protocol for simultaneously performing model selection (hyperparameter tuning) and unbiased performance estimation when data is limited.
- Inner Loop: Used for hyperparameter tuning (using the training fold from the outer loop).
- Outer Loop: Used to assess the performance of the best model found by the inner loop on held-out data.
- When to Use: It is mandatory for providing an unbiased estimate of model performance when you have used the validation set for extensive model development and tuning. It prevents over-optimistic reports of generalizability.
Q6: My dataset is very small (<100 samples). Can I still do a train-validation-test split? A: A traditional three-way split on very small data leads to unreliable estimates due to high variance. The recommended protocol is to use nested cross-validation (as above) or a bootstrap approach.
- Bootstrap Protocol: Repeatedly draw random samples with replacement from your full dataset to create training sets. The out-of-bag samples (those not drawn) form the test set for each iteration. Performance is averaged over many bootstrap iterations. This provides a more stable estimate of performance and confidence intervals on small data.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Rigorous Data Partitioning & Model Evaluation
| Item/Software | Function | Key Consideration |
|---|---|---|
| scikit-learn (Python) | Primary library for train_test_split, StratifiedKFold, GroupShuffleSplit, cross_val_score. |
Ensure version >0.24 for advanced splitting functions. |
| MLxtend (Python) | Provides PredefinedHoldoutSplit and other utilities for implementing rigorous nested CV workflows. |
Useful for enforcing fixed validation sets within CV loops. |
| Pandas DataFrame | Data structure for holding features, targets, and crucial group IDs (patient, batch). | Essential for grouping and stratifying operations. |
| Random State Seed | An integer used to initialize the pseudo-random number generator. | Fixes the reproducibility of your splits. Document this seed. |
| Data Versioning Tool (e.g., DVC, Git LFS) | Tracks exact snapshots of your data and the code that splits it. | Critical for audit trails and reproducible research. |
| Stratification Variable | The array of class labels for classification tasks. | Must be carefully validated for integrity before splitting. |
| Grouping Variable | The array (e.g., Patient_ID) defining non-independent samples. | Must be identified during experimental design. |
Troubleshooting Guides & FAQs
Q1: My model performs excellently during k-fold cross-validation but fails dramatically on the final held-out test set. What went wrong? A: This is a classic symptom of data leakage or improper cross-validation setup. Ensure that all preprocessing steps (e.g., scaling, imputation) are fitted only on the training fold and then applied to the validation fold within each CV loop. Using the entire dataset for preprocessing before splitting biases the estimate. The nested CV protocol is designed to prevent this.
Q2: When using Leave-One-Out Cross-Validation (LOOCV) on my large dataset, the process is computationally prohibitive. What are my options? A: LOOCV fits N models (N = sample size), which is costly. Consider:
- k-Fold with large k: Use 10-fold or 5-fold CV. The variance-bias trade-off is often favorable.
- Stratified k-Fold: If you have a class-imbalanced dataset, use this to preserve class percentages in each fold.
- Efficient Algorithms: For some models (like linear regression), LOOCV can be approximated efficiently via the hat matrix without refitting.
Q3: How do I choose between k-Fold and LOOCV for my small sample size (n<50) study? A: For very small samples, LOOCV provides a nearly unbiased estimate of the true error but can have high variance. Repeated k-Fold CV (e.g., 5-fold repeated 10 times) is often recommended as a good compromise, providing a more stable (lower variance) estimate while mitigating bias from a single random partition.
Q4: I am tuning hyperparameters and selecting features. How do I get a final, unbiased performance estimate for my paper? A: You must use Nested Cross-Validation. An outer loop estimates the generalization error, while an inner loop handles model selection/tuning. Using the same (non-nested) CV for both tuning and performance reporting gives an optimistically biased estimate. See the protocol below.
Q5: My nested CV results show much lower performance than my initial single CV run. Which one should I report? A: Report the nested CV result. The initial, higher estimate was almost certainly biased due to information leakage from the test set into the model selection process. The nested CV result, while perhaps disappointing, is the rigorous, unbiased estimate required for robust method development and publication.
Experimental Protocols & Data Presentation
Protocol 1: Standard k-Fold Cross-Validation
- Randomly shuffle the dataset and split it into k roughly equal-sized, independent folds.
- For each unique fold
i:- Set fold
ias the validation set. - Train the model on the remaining k-1 folds.
- Evaluate the model on the validation fold
iand record the performance metric (e.g., R², RMSE).
- Set fold
- Calculate the final performance estimate as the average of the k recorded metrics.
Protocol 2: Nested Cross-Validation for Unbiased Estimation
- Define an outer loop (e.g., 5-fold or 10-fold) for performance estimation.
- Define an inner loop (e.g., 5-fold) for hyperparameter tuning/model selection.
- For each outer fold
i:- Split data into outer training and outer test sets based on fold
i. - For each candidate hyperparameter set:
- Perform k-fold CV (the inner loop) only on the outer training set.
- Calculate the average performance across inner folds.
- Select the hyperparameter set with the best average inner-loop performance.
- Retrain a model with these optimal parameters on the entire outer training set.
- Evaluate this final model on the held-out outer test set. Record this metric.
- Split data into outer training and outer test sets based on fold
- The final, unbiased performance estimate is the average of the metrics from all outer test sets. The model selection process is thus repeated from scratch for each outer fold, with no leakage.
Performance Comparison of CV Strategies (Simulated Data)
Table 1: Characteristics and typical performance estimates of different cross-validation strategies on a simulated dataset with known true error (0.50). Nested CV provides the most realistic estimate.
| Method | Key Advantage | Key Disadvantage | Estimated Error (Mean ± Std Dev)* | Bias Relative to True Error |
|---|---|---|---|---|
| Hold-Out (70/30) | Computationally cheap | High variance, depends on single split | 0.48 ± 0.05 | Moderate |
| 5-Fold CV | Good bias-variance trade-off | Moderate computational cost | 0.47 ± 0.03 | Low-Moderate |
| 10-Fold CV | Lower bias | Higher computational cost | 0.49 ± 0.02 | Low |
| LOOCV | Low bias, deterministic | Very high variance & cost for large N | 0.50 ± 0.04 | Very Low |
| Nested 5x5 CV | Unbiased for model selection | High computational cost | 0.51 ± 0.02 | Negligible |
*Standard deviation indicates the variance of the estimate across multiple CV runs with different random seeds.
Visualization of Workflows
Title: k-Fold Cross-Validation Workflow
Title: Nested Cross-Validation for Unbiased Estimation
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential computational tools for robust cross-validation and combating overfitting in method development.
| Item | Function in Experiment | Example (Open Source) | Example (Commercial) |
|---|---|---|---|
| ML Framework | Provides core algorithms and CV splitting utilities. | Scikit-learn (Python), caret (R) | SAS JMP, MATLAB Statistics |
| Hyperparameter Optimization Library | Automates search for optimal model parameters within inner CV loop. | Optuna, Hyperopt | SAS Visual Data Mining, H2O.ai |
| Pipeline Tool | Ensures preprocessing steps are correctly contained within each CV fold to prevent data leakage. | Scikit-learn Pipeline |
RapidMiner, Alteryx |
| Stratified Sampling Module | Creates folds that preserve the percentage of sample classes, crucial for imbalanced data. | StratifiedKFold (Scikit-learn) |
Built into most commercial suites |
| High-Performance Computing (HPC) / Cloud Credits | Enables practical use of repeated and nested CV, which are computationally intensive. | SLURM cluster, Google Colab Pro | AWS SageMaker, Azure ML |
| Version Control System | Tracks exact code, parameters, and data splits to ensure full reproducibility of the CV protocol. | Git, DVC | GitHub, GitLab |
Technical Support Center: Troubleshooting & FAQs
Context: This guide supports thesis research focused on amending overfitting in method development and evaluation, specifically when applying regularization to high-dimensional biological data.
Troubleshooting Guides
Issue 1: Model Unstable or Fails to Converge with Genomic Data
- Symptoms: Coefficients vary wildly between runs; warning messages about convergence.
- Probable Cause: High correlation (multicollinearity) among features (e.g., gene expressions from the same pathway); poorly scaled data.
- Solution:
- Standardize Input Features: Always center (mean=0) and scale (variance=1) each predictor independently of the test set. Use training set parameters to transform the test set.
- Increase Regularization Strength: Systematically increase
alpha(λ) parameter. - Try Elastic Net: Set
l1_ratiobetween 0.2 and 0.8 to balance Ridge and LASSO stability. - Check for Constant Features: Remove features with zero variance in the training set.
Issue 2: LASSO Selects Too Many or Too Few Features
- Symptoms: Model is still too complex or overly simplistic, hindering biological interpretation.
- Probable Cause: Suboptimal regularization penalty (
alpha). - Solution:
- Implement Nested Cross-Validation: Use an outer CV for error estimation and an inner CV for
alphaselection to prevent data leakage and overfitting. - Use Information Criterion Paths: For faster search, plot AICc or BIC across a range of
alphavalues and choose the minimum. - Leverage Stability Selection: Run LASSO multiple times on data subsamples and select features that appear consistently.
- Implement Nested Cross-Validation: Use an outer CV for error estimation and an inner CV for
Issue 3: Poor Predictive Performance on Independent Test Set
- Symptoms: High training ( R^2 ), but very low test ( R^2 ) or AUC.
- Probable Cause: Data leakage during preprocessing (e.g., scaling on entire dataset before train/test split) or target variable leakage.
- Solution:
- Audit Preprocessing Pipeline: Ensure all steps (imputation, scaling, feature selection) are fit only on the training fold within each CV loop.
- Validate with External Cohort: Test final locked model on a completely independent, biologically validated dataset.
- Review Feature Origins: Ensure no features are direct proxies for the outcome (e.g., a protein measurement that is part of the clinical diagnostic score).
Frequently Asked Questions (FAQs)
Q1: For genomic data with ~20,000 genes and ~100 samples, should I use Ridge, LASSO, or Elastic Net?
A: Start with Elastic Net. It combines the strengths of both: Ridge regression handles multicollinearity well, while LASSO performs feature selection. Elastic Net's hybrid penalty is particularly effective for ( p >> n ) problems, where it can select more than n features if they are correlated, unlike LASSO. This is common in genomics.
Q2: How do I choose the optimal alpha (λ) and, for Elastic Net, the l1_ratio?
A: Use a search grid with cross-validation.
- Define a log-spaced range for
alpha(e.g., from1e-5to1e2). - For Elastic Net, define a grid for
l1_ratio(e.g.,[0.1, 0.5, 0.7, 0.9, 0.95, 1]). - Perform
GridSearchCVorRandomizedSearchCVusing the appropriate metric (e.g., mean squared error for regression, AUC-ROC for classification). - Crucially: This search must be embedded within a nested cross-validation scheme to obtain unbiased performance estimates for your thesis.
Q3: My features have different scales (e.g., gene expression counts, pH, temperature). Is preprocessing mandatory? A: Yes, it is critical. Regularization penalties are sensitive to feature scale. A feature with larger magnitude will disproportionately influence the penalty term, unfairly shrinking smaller-scale features. Always standardize features to mean=0 and variance=1 based on the training set.
Q4: How do I interpret the coefficients from a regularized model for biological insight? A: Regularized coefficients are shrunken and should be interpreted with caution.
- Non-zero Coefficients (LASSO/Elastic Net): Indicate features selected as important by the model under the given penalty. Pathway enrichment analysis on these selected genes/proteins is a common next step.
- Coefficient Magnitude: While larger absolute values suggest greater influence, they are not directly analogous to effect sizes from ordinary least squares. Focus on the sign (positive/negative association) and the stability of selection across cross-validation folds or bootstrap samples.
- Always validate top selected features with orthogonal experimental methods (e.g., siRNA knockout, ELISA).
Table 1: Core Properties and Application Guidance
| Property | Ridge Regression (L2) | LASSO Regression (L1) | Elastic Net (L1 + L2) |
|---|---|---|---|
| Penalty Term | ( \lambda \sum{j=1}^{p} \betaj^2 ) | ( \lambda \sum{j=1}^{p} |\betaj| ) | ( \lambda \left[ \frac{1-\alpha}{2}\sum{j=1}^{p}\betaj^2 + \alpha \sum{j=1}^{p}|\betaj| \right] ) |
| Feature Selection | No (shinks coefficients) | Yes (can zero out coefficients) | Yes |
| Handles Multicollinearity | Excellent | Poor (selects one) | Good |
| Best For | Dense solutions, many small effects | Sparse solutions, interpretability | High-dim data (p>>n), correlated features |
| Key Hyperparameter | Lambda (alpha) |
Lambda (alpha) |
Lambda (alpha) & L1 Ratio (l1_ratio) |
Table 2: Typical Performance on High-Dimensional Biological Data (Thesis Context)
| Metric | Ridge | LASSO | Elastic Net | Notes for Evaluation Research |
|---|---|---|---|---|
| Avg. Test MSE (Simulated p=1000, n=100) | 0.85 | 0.72 | 0.68 | Elastic Net often shows lower prediction error. |
| Avg. Features Selected | 1000 (all) | 15-30 | 40-80 | LASSO overly aggressive; EN provides more stable biomarker list. |
| Coefficient Bias | Lower | Higher | Medium | Consider bias-variance trade-off in your thesis analysis. |
| Stability (by Bootstrap) | Very High | Low | High | Essential for reproducible method development. |
| Computational Speed | Fast | Fast (with LARS) | Moderate | For p in the 10,000s, use coordinate descent algorithms. |
Standardized Experimental Protocol for Comparative Analysis
Title: Protocol for Comparative Evaluation of Regularization Techniques in Omics Prediction Tasks.
Objective: To empirically evaluate and compare Ridge, LASSO, and Elastic Net regression in preventing overfitting within a high-dimensional omics dataset.
Materials: See "Research Reagent Solutions" below.
Procedure:
- Data Partitioning: Split dataset into independent Discovery (80%) and Hold-out Validation (20%) cohorts. Do not revisit the hold-out set until final model selection is complete.
- Preprocessing (on Discovery Set only):
- Perform missing value imputation using k-NN imputation.
- Standardization: For each feature, subtract the training mean and divide by the training standard deviation. Store these parameters.
- Apply the same transformation to the hold-out set using the stored parameters.
- Hyperparameter Tuning via Nested Cross-Validation:
- On the Discovery set, implement 5x5 Nested CV.
- Outer Loop (5-fold): Defines training/validation splits for performance estimation.
- Inner Loop (5-fold): For each outer training fold, perform a grid search to find optimal hyperparameters (
alphafor Ridge/LASSO;alpha+l1_ratiofor Elastic Net). Use mean squared error as the scoring metric.
- Model Training & Assessment:
- Train a final model on the entire Discovery set using the best hyperparameters found by the inner CV search.
- Evaluate this final model on the locked Hold-out Validation cohort. Report ( R^2 ), MSE, and, for classification, AUC-ROC with 95% CI.
- Stability & Interpretability Analysis:
- Perform 1000 bootstrap resamples on the Discovery set, applying the selected model.
- Record the frequency of feature selection (for LASSO/EN) and coefficient sign stability.
- Perform pathway enrichment analysis (e.g., via g:Profiler, Enrichr) on the consensus feature set.
Visualizations
Diagram 1: Nested CV for Unbiased Evaluation
Title: Nested Cross-Validation Workflow
Diagram 2: Regularization Paths Comparison
Title: Coefficient Paths: LASSO vs. Ridge vs. Elastic Net
Diagram 3: Protocol for Preventing Overfitting
Title: Thesis Anti-Overfitting Protocol with Regularization
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools & Packages for Implementation
| Item/Category | Specific Solution/Software/Package | Function in the Experiment |
|---|---|---|
| Programming Language | Python (≥3.8) with scikit-learn, NumPy, pandas | Core environment for data manipulation, modeling, and analysis. |
| Regularization Algorithms | sklearn.linear_model (Ridge, Lasso, ElasticNet, LassoCV, ElasticNetCV) |
Provides optimized, production-grade implementations of all three techniques. |
| Hyperparameter Tuning | sklearn.model_selection (GridSearchCV, RandomizedSearchCV) |
Essential for automated, rigorous search of alpha and l1_ratio. |
| High-Performance Solver | sklearn.linear_model with coordinate_descent solver |
Efficiently handles datasets where p (features) >> n (samples). |
| Preprocessing | sklearn.preprocessing (StandardScaler) |
Correctly standardizes features to prevent scale bias in penalties. |
| Data Handling | pandas DataFrames |
Manages sample IDs, feature names, and clinical metadata. |
| Visualization | matplotlib, seaborn |
Creates coefficient paths, performance plots, and validation figures. |
| Pathway Analysis | g:Profiler, Enrichr (web) or gseapy (Python) |
Interprets selected gene/protein lists in a biological context. |
| Statistical Validation | scipy.stats (for bootstrapping CIs) |
Quantifies uncertainty in performance metrics and feature stability. |
Principled Feature Selection and Dimensionality Reduction (PCA, PLS) Before Model Fitting
Technical Support Center
Troubleshooting Guides & FAQs
Q1: My PCA model yields unstable loadings between runs, causing irreproducible feature selection. What are the causes and solutions? A: This is often caused by (a) features with vastly different scales or (b) near-eigenvalues leading to arbitrary axis rotations.
- Solution 1: Always standardize (center and scale to unit variance) each feature before applying PCA. This ensures all features contribute equally to the variance.
- Solution 2: If instability persists, check eigenvalue gaps. Use a stricter tolerance for eigenvalue equality or consider using bootstrapping to assess loading stability. Select features with consistently high absolute loadings across bootstrap samples.
Q2: How do I choose the optimal number of components for PCA or PLS to avoid overfitting in my predictive model? A: The goal is to retain signal and discard noise. Do not rely solely on variance explained.
- For PCA (unsupervised): Use cross-validation on the downstream model. Perform PCA with varying
n_componentswithin each CV fold, train the model on the transformed training set, and evaluate on the transformed validation set. Choosen_that minimizes validation error. - For PLS (supervised): Use built-in CV methods like
SIMPLSorNIPALSwith a defined number of latent variables. Employ criteria like the first local minimum in Prediction Residual Sum of Squares (PRESS) plot or a 1-standard-error rule.
Q3: After PLS, my model still overfits despite using latent variables. What went wrong? A: PLS is not immune to overfitting, especially with small sample sizes (n) and very large feature counts (p).
- Cause: PLS maximizes covariance, which can still fit noise. With small
n, the estimated latent directions may be spurious. - Solution: Implement double cross-validation: an outer loop for generalizability estimation and an inner loop for rigorous optimization of the number of PLS components. Also, consider
sparse PLSmethods that perform feature selection within the PLS framework to reduce noise.
Q4: I have missing data in my dataset. Can I apply PCA/PLS, and what are the principled methods to handle it? A: Standard PCA/PLS requires a complete matrix. Imputation is necessary but must be done cautiously to prevent bias.
- Recommended Protocol:
- Perform initial filtering: Remove features with excessive (>20%) missingness if not biologically critical.
- Use model-based imputation: Apply Multivariate Imputation by Chained Equations (MICE) or matrix completion algorithms, iterating over the feature columns. For omics data, consider
k-nearest neighbors impute using similar samples. - Post-imputation validation: Conduct PCA on the imputed data and color samples by their original missingness pattern to check for introduced artifacts.
Q5: How do I interpret selected features from PCA/PLS in the context of biological mechanism or drug response? A: Projection methods provide transformed components, not direct feature selection.
- For PCA: Examine the
loadings(coefficients) of the top PCs. Features with large absolute loadings (positive or negative) drive that component's variation. Biologically interpret the collective meaning of these co-varying features. - For PLS: Examine the
weightsorVIP(Variable Importance in Projection) scores. Features with high VIP (>1.0) are most relevant for predicting the response. Validate these against known pathways or through independent literature mining.
Table 1: Comparison of Dimensionality Reduction Methods in Overfitting Context
| Method | Supervised? | Primary Objective | Feature Selection Direct? | Overfitting Risk (Small n, Large p) | Key Hyperparameter |
|---|---|---|---|---|---|
| PCA | No | Maximize variance in X | No (but via loading thresholds) | Moderate (no Y guidance) | Number of Components |
| PLS | Yes | Maximize covariance(X, Y) | No (but via VIP scores) | High if components not validated | Number of Latent Variables |
| Sparse PCA | No | Maximize variance with L1 penalty | Yes (loadings forced to zero) | Lower than PCA | Sparsity Parameter (Alpha) |
| Sparse PLS | Yes | Maximize covariance with L1 penalty | Yes (weights forced to zero) | Lower than standard PLS | Sparsity Parameter (Eta) |
Table 2: Typical Results from a PCA Cross-Validation Experiment to Determine Optimal Components
| Number of PCs Retained | Cumulative Variance Explained (%) | Mean CV RMSE of Downstream Model | Std Dev of CV RMSE |
|---|---|---|---|
| 2 | 45.2 | 1.85 | 0.32 |
| 5 | 72.8 | 1.21 | 0.18 |
| 8 | 88.5 | 0.98 | 0.21 |
| 10 | 92.1 | 1.05 | 0.25 |
| 15 | 97.3 | 1.34 | 0.41 |
Experimental Protocols
Protocol 1: Cross-Validated PCA for Regression Model (To Prevent Overfitting)
- Preprocessing: Split full dataset
Dinto independent Training/Test sets (e.g., 80/20). Work only on the Training setT. - Standardization: On
T, center and scale each feature to unit variance. Store the mean and standard deviation. - Nested CV Loop: For each fold in
k-fold CV: a. SplitTinto training (Tr) and validation (Val) subsets. b. Apply standardization toTrusing its parameters, transformValwith same. c. Forcin [1 toC_max] (e.g., 1 to 20): i. Fit PCA withccomponents onTr. ii. TransformTrandValtoTr_pc,Val_pc. iii. Train your regression model (e.g., Ridge Regression) onTr_pc. iv. Predict onVal_pcand record error. - Determine Optimal
c: Average validation error across folds for eachc. Choosec_optthat gives minimum average error. - Final Model: Fit PCA with
c_opton entireT, transformT. Train final model. Apply stored standardization and PCA transformation to the held-out Test set for final unbiased evaluation.
Protocol 2: VIP-based Feature Selection after PLS (For Interpretable Biomarker Discovery)
- Data Preparation: Use standardized matrix
Xand response vectorY. - Initial PLS Model: Fit a PLS regression model with a generous number of latent variables (LVs), e.g., 10.
- Component Selection: Plot cross-validated RMSE vs. number of LVs. Select the number
l_optat the first clear minimum. - Refit Model: Refit PLS with exactly
l_optLVs on the full training set. - Calculate VIP Scores: For each feature
j, calculate VIP = sqrt( p * Σ(SScontribl * (weight{lj}^2)) / Σ(SScontrib_l) ), where summation is overl_optLVs,pis total features,SS_contrib_lis the sum of squares explained by the LVl, andweight_{lj}is the PLS weight. - Threshold Features: Retain features with VIP > 1.0 as important for the model. Validate this subset in an independent test set or via pathway enrichment analysis.
Diagrams
Title: Workflow for Principled Dimensionality Reduction in Model Building
Title: Nested CV for PCA Component Selection
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Computational Tools for Feature Selection & Dimensionality Reduction
| Item (Software/Package) | Category | Primary Function | Key Consideration for Overfitting |
|---|---|---|---|
| scikit-learn (Python) | Core Library | Provides PCA, PLSRegression, cross_val_score, GridSearchCV. |
Ensures correct CV separation; pipelines prevent data leakage. |
| mixOmics (R/Bioc) | Omics-Focused | Implements sparse PLS, sPCA, VIP calculation, DIABLO for multi-omics. |
Designed for high-dimensional biological data with built-in CV. |
| SIMCA-P+ (Commercial) | Standalone Software | Industry-standard for multivariate analysis (PCA, PLS, OPLS). | Uses sophisticated metrics (R2X, R2Y, Q2) to guide component choice. |
| MissForest (R) / IterativeImputer (Python) | Data Imputation | Advanced model-based imputation for missing data. | Reduces bias in pre-PCA/PLS data preparation compared to mean impute. |
| MATLAB Statistics & ML Toolbox | Computational Environment | Comprehensive suite for matrix computation and chemometrics. | Offers detailed diagnostic plots (e.g., scores, loadings, residuals). |
| VIPER (R Package) | Visualization | Creates Variable Importance in PLS Projection (VIP) plots. | Aids in objective, visual thresholding of important features. |
Incorporating Domain Knowledge and Biological Constraints to Guide Model Building
Technical Support Center: Troubleshooting Guides & FAQs
FAQ 1: How can biological plausibility constraints be practically enforced in a neural network model to prevent overfitting to noisy in vitro data? Answer: Implement custom penalty terms or architectural constraints. For example, use a pathway-constrained layer where neuron connections mirror a known signaling pathway (e.g., EGFR-RAS-MAPK). Connections not present in the canonical pathway are forced to zero, drastically reducing spurious parameters. A recent study (2023) showed this reduced overfitting (lower test set MSE) by 40% compared to an unconstrained Dense Neural Network (DNN) on high-content screening data.
FAQ 2: My model trained on cell line data fails to generalize to patient-derived organoids. What domain knowledge can guide adaptation? Answer: This is a classic domain shift issue. Incorporate knowledge of the tumor microenvironment (TME). Create a knowledge graph of TME components (e.g., fibroblasts, immune cells, ECM) and their known interactions with tumor cells. Use this graph to structure a multi-modal input layer or to generate synthetic training samples that simulate TME influences, moving beyond cell-line monoculture data.
FAQ 3: When using genomics data for drug response prediction, how do I prevent the model from latching onto technical batch effects instead of real biological signals? Answer: Integrate known batch covariates (sequencing platform, lab) directly as invariant features. Employ a Domain Adversarial Neural Network (DANN) where a primary network learns predictive features, and an adversarial discriminator tries to predict the batch from those features. The gradient is reversed during training, forcing the primary network to learn batch-invariant, biologically relevant representations.
FAQ 4: How can I incorporate known physical or thermodynamic constraints (e.g., binding energy limits) into a predictive model for protein-ligand affinity? Answer: Use the constraints as soft bounds via loss function penalties or as hard bounds via activation functions. For instance, scale the final layer's output with a sigmoid function bounded by the known theoretical maximum binding affinity. A 2024 benchmark showed that such physically-constrained models improved prediction robustness on novel scaffold compounds by 25% in terms of RMSE.
Table 1: Comparison of Constraint Methods for Preventing Overfitting
| Constraint Type | Enforcement Method | Typical Use Case | Reported Reduction in Test Error* |
|---|---|---|---|
| Pathway Topology | Sparse, Masked Layers | Signaling Response Prediction | 35-45% |
| Physical Bounds | Output Layer Activation | Binding Affinity Prediction | 20-30% |
| Invariance (e.g., Batch) | Adversarial Training | Multi-study Genomics | 40-60% |
| Causal Structure | Graph-Guided Architecture | Drug Synergy Prediction | 30-50% |
Compared to an equivalent unconstrained model on held-out experimental validation sets. (Synthetic summary of recent literature, 2023-2024)
Experimental Protocols
Protocol: Evaluating a Pathway-Constrained Neural Network
Objective: To assess if enforcing a known signaling pathway topology reduces overfitting in a dose-response prediction task.
- Data Preparation: Use a phosphoproteomics dataset (e.g., LINCS L1000) measuring protein phosphorylation under drug perturbations.
- Model Definition:
- Control Model: A standard fully-connected DNN.
- Constrained Model: A DNN where the first hidden layer's weight matrix is element-wise multiplied by a binary adjacency matrix A representing a canonical pathway (e.g., from KEGG or Reactome).
- Training: Split data into training (60%), validation (20%), test (20%). Train both models to minimize MSE on training data. Use early stopping on validation loss.
- Evaluation: Compare the models on the held-out test set using MSE and R². Perform a permutation test on input features to measure the specificity of the constrained model to pathway-relevant inputs.
Protocol: Adversarial Batch Effect Removal (DANN)
Objective: To learn gene expression features invariant to technical batch for robust biomarker discovery.
- Data: Collect gene expression data from multiple public studies (e.g., TCGA, GEO) for a disease state, ensuring batch labels are known.
- Network Architecture:
- Feature Extractor (G): Shared layers mapping input gene expression to a latent feature space.
- Label Predictor (L): Takes features from G to predict disease subtype/outcome.
- Batch Discriminator (D): Takes features from G to predict the batch label.
- Training: Use a combined loss:
Loss = Loss_label(G, L) - λ * Loss_batch(G, D). The negative sign on the batch loss implements gradient reversal during backpropagation, encouraging G to learn features that confuse D. - Validation: Evaluate the label predictor L on a completely held-out study (a new batch) to measure cross-study generalizability.
Visualizations
Diagram 1: Neural Network with Pathway Topology Constraints
Diagram 2: Domain Adversarial Neural Network for Batch Removal
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Constrained Model Development & Validation
| Item / Reagent | Provider Examples | Function in Context |
|---|---|---|
| LINCS L1000 Data | NIH LINCS Program | Large-scale perturbational transcriptomics dataset for training and testing models on cell response. |
| KEGG/Reactome Pathway Maps | Kanehisa Labs / EMBL-EBI | Source of canonical pathway adjacency matrices used to constrain model architectures. |
| Cell Signaling Multiplex Assays | Luminex, MSD, IsoPlexis | Generate high-dimensional protein activity data for validating model predictions on constrained pathways. |
| Patient-Derived Organoid (PDO) Models | Commercial Biobanks (e.g., CrownBio) | Gold-standard ex vivo system for testing model generalizability beyond cell lines. |
| Domain Adversarial Training Code | GitHub (e.g., DANN-PyTorch) | Open-source implementations of adversarial de-confounding algorithms. |
| Physics-Informed NN Libraries | PyTorch, TensorFlow with custom layers | Frameworks for implementing hard/soft physical constraints as part of the model loss. |
Setting a Priori Hypotheses and Analysis Plans to Minimize Researcher Degrees of Freedom
Troubleshooting Guides and FAQs
FAQ 1: What constitutes a valid a priori hypothesis versus a post hoc explanation? A valid a priori hypothesis must be specified before data collection or analysis begins. It includes a clear statement of the relationship between variables, the direction of the effect, and the specific statistical test to be used. Post hoc explanations are generated after seeing the data and are considered exploratory; they require independent validation and should not be reported with the same statistical confidence.
FAQ 2: How specific should my analysis plan be to prevent unintentional p-hacking? Your plan must unambiguously define: primary and secondary endpoints, exact model specifications (including covariates), data handling rules for outliers/missing data, the precise statistical test, and the alpha level for significance. Ambiguity in any of these areas creates researcher degrees of freedom.
FAQ 3: My experiment yielded an unexpected but promising result. How should I proceed? Document the unexpected finding clearly as exploratory. Do not present it as a confirmatory test. The finding must then be used to generate a new, specific a priori hypothesis for a future, preregistered experiment designed explicitly to test it.
FAQ 4: What are the key elements of a preregistration document for a preclinical study? Key elements include: detailed experimental protocol (species, sample size justification, randomization method), blinding procedures, primary outcome measure and how it is quantified, statistical analysis plan, and criteria for excluding data. Platforms like the Open Science Framework (OSF) or preclinical trial registries provide templates.
FAQ 5: How do I handle necessary protocol deviations without compromising the analysis plan? All deviations must be documented in real-time. The pre-specified analysis should be run on the intent-to-treat dataset. A sensitivity analysis can then be conducted to assess the impact of the deviation, but the primary result comes from the planned analysis on all collected data.
Troubleshooting Guide: Common Issues and Solutions
| Issue | Symptoms | Root Cause | Solution |
|---|---|---|---|
| Low Statistical Power | Non-significant result despite large effect trend. | Underpowered design due to small sample size (N). | Pre-experiment: Use power analysis to determine N. Post-experiment: Report effect size with confidence interval; avoid "accepting the null." |
| Model Overfitting | Excellent model fit in training data, fails in validation. | Too many model parameters/predictors relative to observations. | Preregister model; use hold-out validation samples; apply regularization techniques (e.g., ridge regression). |
| Inconsistent Blinding | Effect sizes are larger in non-blinded assessments. | Confirmation bias influencing subjective measurements. | Automate data capture where possible; use third-party coding; document blinding protocol failures. |
| Multiple Testing Inflation | Several secondary outcomes are "just significant" (p ~ 0.05). | Conducting many statistical tests without correction. | Pre-specify one primary outcome; for secondary analyses, use corrected alpha (e.g., Bonferroni, FDR). |
| Selective Reporting | Only "successful" experiments or endpoints are published. | File drawer problem; publication bias. | Preregister all studies; report all pre-specified outcomes regardless of result; publish negative findings. |
Key Experimental Protocols
Protocol 1: Preregistration and Blinded Analysis Workflow
- Design Phase: Formulate primary hypothesis and select primary endpoint. Conduct formal sample size/power calculation.
- Preregistration: Document steps 1, 3, 4, 5, and 6 on a timestamped, immutable registry (e.g., OSF).
- Experimental Execution: Randomize subjects, implement blinding, collect data according to protocol.
- Data Freeze: Lock raw data file upon completion. Create a copy for analysis.
- Blinded Analysis: With outcome data blinded (e.g., coded as Group A/B), run the pre-specified analysis script to generate results.
- Unblinding & Interpretation: Unblind groups and interpret the results of the pre-run analysis.
Protocol 2: Hold-Out Validation for Predictive Models
- Pre-Data Collection: Randomly assign future subjects to a "training" set (e.g., 70%) and a "hold-out validation" set (30%). This assignment is preregistered.
- Model Development: Collect data for the training set. Develop the model (e.g., biomarker signature, machine learning algorithm) using only this data.
- Model Lock: Finalize all model parameters and thresholds. Do not modify after seeing validation data.
- Validation: Collect data for the hold-out set. Apply the locked model from step 3. Assess performance (e.g., accuracy, AUC) on this fresh data.
Table 1: Impact of Researcher Degrees of Freedom on False-Positive Rates
| Analysis Practice | Nominal Alpha | Estimated False-Positive Rate | Key Reference |
|---|---|---|---|
| Single test, pre-specified | 0.05 | 0.05 | Simmons et al., 2011 |
| Testing multiple endpoints | 0.05 | 0.23 (for 4 outcomes) | Althouse, 2016 |
| Optional stopping (peeking at data) | 0.05 | >0.20 | Lakens, 2014 |
| Choosing covariates post-hoc | 0.05 | 0.29 | Simmons et al., 2011 |
| Preregistration + blinded analysis | 0.05 | ~0.05 | Nosek et al., 2018 |
Table 2: Recommended Sample Sizes for Common Preclinical Designs (Power=0.8, Alpha=0.05)
| Experimental Design | Effect Size (Cohen's d) | Required N per Group | Notes |
|---|---|---|---|
| Two-group comparison (t-test) | Large (0.8) | 26 | Common for pilot studies. |
| Two-group comparison (t-test) | Medium (0.5) | 64 | Adequate for many interventions. |
| Two-group comparison (t-test) | Small (0.2) | 394 | Often impractical in preclinical work. |
| One-way ANOVA (3 groups) | Medium (f=0.25) | 52 per group (156 total) | For comparing multiple treatments. |
Visualizations
Title: Preregistered Experiment Workflow
Title: Causes of Overfitting and Solutions
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Minimizing Degrees of Freedom |
|---|---|
| Preregistration Template (OSF/AsPredicted) | Provides a structured format to document hypotheses, methods, and analysis plans before data collection, creating an immutable record. |
| Randomization Software (e.g., GraphPad QuickCalcs) | Generates unpredictable allocation sequences to eliminate selection bias, a key component of blinding. |
| Statistical Power Calculator (G*Power) | Allows for formal sample size justification based on expected effect size and desired power, reducing underpowered studies. |
| Electronic Lab Notebook (ELN) | Timestamps all raw data entries and protocol deviations, providing an auditable trail for replication and peer review. |
| Blinding/Coding Scripts (R/Python) | Scripts to anonymize group allocation during data analysis, preventing conscious or unconscious bias during statistical testing. |
| Data Visualization Tool (Pre-spec Charts) | Templates for pre-specified data visualizations (e.g., bar graphs with 95% CIs) that are populated with data, preventing "plot shopping." |
| Registered Reports Format | A publishing format where the introduction and methods are peer-reviewed before results are known, aligning incentives with rigorous methods. |
Diagnosing the Problem: How to Detect and Correct an Overfit Model
Troubleshooting Guides & FAQs
Q1: My model performs exceptionally well on the training set but poorly on the validation set. What is the primary cause, and what are the first diagnostic steps? A1: This is the classic signature of overfitting. The model has learned patterns specific to the training data, including noise, that do not generalize. First steps:
- Compare Learning Curves: Plot training and validation loss/accuracy per epoch.
- Check Data Fidelity: Ensure no data leakage between sets and that the validation set is representative.
- Simplify the Model: Reduce model capacity (e.g., fewer layers/neurons) and retrain.
Q2: During training, validation loss decreases initially but then starts to increase while training loss continues to decrease. What does this mean, and how do I address it? A2: This indicates the onset of overfitting after a certain number of epochs. You should implement Early Stopping. Halt training when the validation loss fails to improve for a pre-defined number of epochs (patience). The best model is the one saved at the epoch with the lowest validation loss.
Q3: What are the most effective regularization techniques to narrow the performance gap for a deep neural network in image-based assay analysis? A3: A multi-pronged regularization approach is best:
- L1/L2 Weight Regularization: Penalizes large weights in the loss function.
- Dropout: Randomly deactivates a fraction of neurons during training.
- Data Augmentation: Artificially expands the training set with label-preserving transformations (e.g., rotation, flip, noise addition for microscopy images).
- Batch Normalization: Stabilizes layer inputs, allowing for higher learning rates and acting as a mild regularizer.
Q4: How can I determine if my train/validation split is biased or unrepresentative, contributing to the gap? A4: Perform Stratified K-Fold Cross-Validation. If the model performance varies wildly across different folds, your initial split was likely unlucky or your data has underlying subgroups not proportionally represented. Cross-validation provides a more robust performance estimate.
Q5: For a random forest model predicting compound activity, the training accuracy is ~95% but validation is ~70%. Does this imply overfitting, and what hyperparameters should I tune first? A5: Yes, this suggests overfitting. Key random forest hyperparameters to regularize the model:
- Increase
min_samples_leaf(minimum samples required to be at a leaf node). - Increase
min_samples_split(minimum samples required to split an internal node). - Reduce
max_depth(maximum depth of the tree). - Increase
max_features(number of features to consider for the best split).
| Symptom | Likely Cause | Primary Diagnostic Action | Common Solution |
|---|---|---|---|
| High train accuracy, low val accuracy | Overfitting | Plot learning curves | Increase regularization, get more data |
| Validation loss increases after a point | Overfitting (no early stopping) | Monitor val loss per epoch | Implement early stopping callback |
| High variance in metrics across folds | Unrepresentative data split or small dataset | Perform k-fold cross-validation | Use stratified splitting, collect more data |
| Poor performance on both sets | Underfitting or data mismatch | Check model architecture & data preprocessing | Increase model capacity, check feature quality |
| Metrics jump dramatically on new test set | Data leakage or non-i.i.d. data | Audit data splitting & preprocessing pipeline | Ensure no target information leaks, re-split |
Experimental Protocol: Diagnosing Overfitting with Learning Curves
Objective: To visually diagnose overfitting by comparing model performance on training and validation sets across training epochs.
Materials: See "Research Reagent Solutions" table.
Methodology:
- Data Preparation: Split the dataset into three independent sets: Training (70%), Validation (15%), and Hold-out Test (15%). Ensure stratification for classification tasks.
- Model Compilation: Initialize your model (e.g., a CNN for image data). Use a standard optimizer (Adam) and loss function.
- Training with History Logging: Train the model for a generous number of epochs (e.g., 100), setting the
validation_dataparameter to the validation set. Ensure the training function returns a history object. - Data Extraction: From the history object, extract lists of: training loss, validation loss, training metric (e.g., accuracy), validation metric per epoch.
- Visualization: Generate two side-by-side plots:
- Plot 1 (Loss): X-axis = Epoch, Y-axis = Loss. Plot training loss and validation loss.
- Plot 2 (Accuracy): X-axis = Epoch, Y-axis = Accuracy. Plot training accuracy and validation accuracy.
- Interpretation: Identify the epoch where validation loss diverges and begins to increase while training loss continues to decrease. This is the optimal early stopping point.
Visualizations
Diagram Title: Learning Curve Analysis Workflow for Overfitting Diagnosis
Diagram Title: Model Capacity Impact on Generalization and Performance Gaps
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Experiment |
|---|---|
| Deep Learning Framework (TensorFlow/PyTorch) | Provides libraries for building, training, and evaluating complex neural network models. |
| scikit-learn | Offers tools for data splitting (traintestsplit), cross-validation, and implementing traditional ML models with regularization. |
| Weights & Biases (W&B) / TensorBoard | Experiment tracking platforms to log, visualize, and compare learning curves and model metrics in real-time. |
| Data Augmentation Pipeline (Albumentations/torchvision) | Systematically generates augmented training samples (rotated, flipped, etc.) to improve generalization. |
| Early Stopping Callback | Automatically halts training when validation performance plateaus or degrades, preventing overfitting. |
| Hyperparameter Optimization Library (Optuna/Ray Tune) | Automates the search for optimal regularization parameters (e.g., dropout rate, weight decay). |
| Stratified K-Fold Splitter | Ensures representative distribution of classes across all training/validation splits, reducing bias. |
Technical Support Center: Troubleshooting Guides & FAQs
Thesis Context: This support content is framed within a broader research thesis focused on addending overfitting in method development and evaluation. Effective stability testing is a critical, yet often overlooked, defense against overfitting, ensuring developed models and methods are robust, generalizable, and not merely tuned to idiosyncrasies of a specific dataset.
Frequently Asked Questions (FAQs)
Q1: Why is stability testing for small perturbations critical in preventing overfitting in predictive modeling for drug discovery? A: Overfitting occurs when a model learns not only the underlying signal but also the noise specific to the training dataset. Small, realistic perturbations (e.g., minor measurement errors, batch effects) represent a form of noise. If a model's predictions or selected features change drastically due to these minor changes, it is a hallmark of overfitting—the model is brittle and likely capturing noise. Stability testing validates that the core findings are data-driven and not artifact-driven, a prerequisite for reliable translational research.
Q2: During cross-validation, my model performance metrics (e.g., R², AUC) vary widely between folds. Is this a stability issue, and how should I address it? A: Yes, high variance in performance across cross-validation folds indicates instability and potential overfitting to specific fold compositions. This suggests your model may be too complex or the dataset is too small. To address this:
- Simplify the Model: Increase regularization strength (e.g., higher lambda in LASSO, alpha in ridge regression).
- Increase Data: Use data augmentation techniques suitable for your domain (e.g., adding noise, SMOTE for limited bioactivity classes) or acquire more data.
- Use Nested CV: Employ nested cross-validation to strictly separate hyperparameter tuning from performance evaluation, providing a more reliable and stable estimate of generalizability.
Q3: My feature selection process yields a completely different list of "important" biomarkers every time I resample the data. What does this mean, and what stable selection methods are recommended? A: This is a classic sign of feature selection instability, severely undermining the interpretability and reproducibility of your research—key concerns in method development. To improve stability:
- Employ Stability Selection: Use methods like randomized LASSO across many bootstrap samples. Features frequently selected across samples are considered stable.
- Use Ensemble Methods: Leverage Random Forests or gradient boosting, which aggregate feature importance across many data subsets.
- Apply Consensus Clustering: For high-dimensional 'omics data, cluster features (genes/proteins) based on correlation before selection, then select stable cluster representatives.
Q4: How can I formally quantify and report the sensitivity of my model to data perturbations? A: You should implement a perturbation analysis and report quantitative stability metrics. A standard protocol is outlined below.
Experimental Protocol: Quantifying Model Stability via Perturbation Analysis
Objective: To quantitatively assess the sensitivity of a trained predictive model to small, random perturbations in the input data.
Materials & Workflow:
Diagram Title: Workflow for Perturbation-Based Stability Testing
Detailed Protocol:
- Baseline Model: Train your model on the original training dataset
D. Evaluate its performance (e.g., AUC, RMSE) on a fixed, pristine holdout test set. Record this asP_baseline. - Perturbation Generation: Create
Mnew training datasets (e.g., M=100). For each:- For continuous features, add Gaussian noise:
X'_i = X + ε, whereε ~ N(0, σ). Setσto a small fraction (e.g., 0.01-0.05) of the feature's standard deviation. - For categorical features or labels, simulate minor label flipping with a very low probability (e.g., 0.01).
- For continuous features, add Gaussian noise:
- Retraining: Train the same model architecture (with identical hyperparameters) on each of the
Mperturbed datasetsD'_i. - Evaluation: Evaluate each of the
Mnewly trained models on the same, original holdout test set from Step 1. Record the performanceP_ifor each model. - Stability Quantification: Calculate the mean (
μ) and standard deviation (σ) of theMperformance scores{P_1, ..., P_M}. A lowσindicates high stability. The degradation fromP_baselinetoμindicates robustness to noise.
Key Research Reagent Solutions & Materials
| Item | Function in Stability Testing |
|---|---|
| Bootstrapped Datasets | Resampled datasets with replacement, used to simulate data variability and assess model/feature selection consistency. |
| Regularization Reagents (L1/L2) | "Penalties" added to the model's loss function (e.g., LASSO, Ridge) to constrain complexity, directly combating overfitting and improving stability. |
| Nested Cross-Validation Loop | An experimental design framework that isolates model tuning from validation, preventing optimistic bias and yielding a more stable performance estimate. |
| Consensus Clustering Algorithm | A tool for identifying stable feature clusters in high-dimensional data, reducing the noise from individual unstable features. |
| Stability Selection Library | Software implementation (e.g., stabs in R, scikit-learn-extra) of the randomized LASSO procedure for stable feature selection. |
Table 1: Example Stability Analysis of Three Classifier Types on a Perturbed Transcriptomics Dataset (Performance Metric: Area Under the ROC Curve (AUC))
| Model Type | Baseline AUC (No Perturbation) | Mean AUC after Perturbation (μ) | Std. Dev. of AUC (σ) | Stability Interpretation |
|---|---|---|---|---|
| Complex Deep Neural Net | 0.95 | 0.87 | 0.08 | Unstable. High performance drop and high variance indicate overfitting to training data noise. |
| Random Forest (Ensemble) | 0.92 | 0.90 | 0.03 | Stable. Minimal performance drop and low variance demonstrate robustness. |
| Logistic Regression (L1) | 0.88 | 0.86 | 0.02 | Very Stable. Lowest variance, though baseline performance is lower. Favors generalizability. |
Table 2: Impact of Regularization Strength on Model Stability
| Regularization Parameter (α) | Mean # of Selected Features | Feature Selection Stability Index* | Test Set RMSE (μ ± σ) |
|---|---|---|---|
| 0.001 (Weak) | 145 | 0.31 | 1.52 ± 0.41 |
| 0.1 (Moderate) | 22 | 0.85 | 1.08 ± 0.09 |
| 1.0 (Strong) | 7 | 0.98 | 1.21 ± 0.05 |
*Stability Index: Jaccard similarity of selected feature sets across 100 bootstrap samples.
Diagram Title: Stability Testing as a Guard Against Overfitting in Research
Utilizing Learning Curves to Diagnose High Variance and Guide Data Collection
Technical Support Center: Troubleshooting Guides and FAQs
FAQ: Core Concepts
Q1: What are learning curves in the context of method development research? A: Learning curves are diagnostic plots that show a model's performance (e.g., error or accuracy) on both training and validation sets as a function of the amount of training data or the number of training iterations. In our thesis on addending overfitting, they are the primary tool to visually diagnose the bias-variance trade-off, specifically identifying when high variance (overfitting) is the limiting factor in model performance.
Q2: How can a learning curve diagnose "High Variance"? A: A classic high variance signature is indicated by a large gap between the training and validation curves. The training error remains low (or accuracy high), while the validation error is significantly higher and plateaus. This gap indicates that the model has memorized the training data noise and structure but fails to generalize to unseen validation data—the definition of overfitting.
Q3: When should I consider collecting more data based on the learning curve? A: Data collection is most effective when the learning curve shows a high variance pattern and the validation curve has not yet fully converged to a plateau. If both curves are converging and continue to decrease (or increase for accuracy) with more data, adding data is likely to improve performance. If the validation curve has flatlined, more data alone may not help, and architectural changes (e.g., increased regularization) are needed first.
Troubleshooting Guide: Common Experimental Issues
Issue 1: Validation error is much higher than training error, and both seem to have plateaued.
- Diagnosis: Severe high variance/overfitting, potentially with a model capacity that is too high for the current data complexity. The plateau suggests more data may not fully resolve the issue.
- Action Plan:
- Increase Regularization: Apply or strengthen techniques like L1/L2 regularization, dropout (for neural networks), or data augmentation.
- Simplify the Model: Reduce the number of parameters (e.g., fewer layers, fewer nodes per layer, shallower trees).
- Feature Engineering: Reduce redundant or irrelevant features.
- Re-evaluate Data: After implementing the above, if the validation curve shows a downward trend again, then consider adding more data.
Issue 2: Both training and validation error are high and close together.
- Diagnosis: High bias (underfitting). The model is too simple to capture the underlying patterns.
- Action Plan:
- Increase Model Complexity: Add more layers, nodes, or polynomial features.
- Reduce Regularization.
- Extend Training Time (for iterative models).
- Engineer more relevant features. Collecting more data is not the primary solution here.
Issue 3: The validation curve is noisy/jumpy.
- Diagnosis: Often due to a validation set that is too small or not properly shuffled/stratified.
- Action Plan:
- Increase the size of your validation set.
- Use k-fold cross-validation to generate more stable learning curve estimates.
- Ensure your data split maintains the same distribution of classes/critical features (stratified split).
Experimental Protocol: Generating a Diagnostic Learning Curve
Objective: To diagnose bias-variance and guide data collection strategy. Methodology:
- Data Preparation: Split your dataset into a fixed Test Set (e.g., 20%, held back for final evaluation only) and a Training Pool (80%).
- Create Subsets: From the Training Pool, create a series of incrementally larger training subsets (e.g., 10%, 20%, 40%, 60%, 80%, 100% of the Training Pool). Use a fixed, separate Validation Set (e.g., 20% of the original data, held out from the Training Pool) for all evaluations.
- Train & Validate: For each training subset:
- Train your model from scratch.
- Calculate the performance metric (e.g., Mean Squared Error, Accuracy) on the training subset itself.
- Calculate the same metric on the fixed Validation Set.
- Plot: Create a plot with dataset size (or % of total) on the x-axis and the performance metric on the y-axis. Plot two lines: Training Score and Validation Score.
- Analyze: Refer to the decision logic in the workflow diagram below.
Visualizations
Diagram 1: Learning Curve Analysis Workflow
Diagram 2: Signature Learning Curve Patterns
Table 1: Impact of Data Size on Model Performance (Hypothetical Case Study)
| Training Data Size (%) | Training MSE | Validation MSE | Gap (Val - Train) | Diagnosis Hint |
|---|---|---|---|---|
| 10 | 0.08 | 0.45 | 0.37 | High Variance |
| 25 | 0.10 | 0.32 | 0.22 | High Variance |
| 50 | 0.12 | 0.23 | 0.11 | High Variance |
| 75 | 0.14 | 0.19 | 0.05 | Improving |
| 100 | 0.15 | 0.18 | 0.03 | Near Ideal |
Table 2: Intervention Efficacy for High Variance Scenarios
| Intervention Applied | Avg. Validation MSE Before | Avg. Validation MSE After | % Improvement | Recommended Data Collection Impact |
|---|---|---|---|---|
| L2 Regularization (λ=0.1) | 0.35 | 0.24 | 31.4% | Higher ROI for new data |
| Dropout (rate=0.5) | 0.35 | 0.22 | 37.1% | Higher ROI for new data |
| Feature Selection (Top 50%) | 0.35 | 0.28 | 20.0% | Moderate ROI for new data |
| None (Baseline) | 0.35 | 0.35 | 0.0% | Low ROI for new data |
The Scientist's Toolkit: Research Reagent Solutions
| Item/Category | Function in Learning Curve Experiments |
|---|---|
| Scikit-learn | Python library providing easy-to-use functions (learning_curve, validation_curve) to generate plot data. |
| TensorBoard / Weights & Biases | Tracking tools to visualize model performance across training runs and dataset sizes automatically. |
Cross-Validation Splitters (KFold, StratifiedKFold) |
Essential for creating robust, non-noisy validation scores when generating learning curve points. |
| Regularization Modules (L1/L2, Dropout Layers) | Direct interventions to apply when high variance is diagnosed, reducing overfitting. |
| Data Augmentation Pipelines | Artificially increases effective training data size and diversity, directly addressing high variance. |
| Synthetic Data Generators | For fields with scarce data, can create preliminary datasets to shape model architecture before costly real data collection. |
Simulation and Permutation Testing to Establish a Baseline for Random Performance
Technical Support Center: Troubleshooting Guides & FAQs
FAQ Context: This support center is designed to assist researchers in implementing robust simulation and permutation testing methods. Proper application of these techniques is critical for establishing a rigorous baseline of random performance, a foundational step in preventing overfitting during the development and evaluation of new analytical methods, models, or biomarkers in drug development.
Frequently Asked Questions
Q1: During a permutation test for a new biomarker signature, my p-value is calculated as 0.000. Is this valid, and what might it indicate? A: A p-value of 0.000 typically means no permuted statistic exceeded your observed statistic in, for example, 10,000 permutations. While statistically significant, it warrants investigation.
- Potential Issue: An error in the permutation procedure, such as incorrectly shuffling labels within batches or groups, failing to preserve the underlying data structure, or an overly simplistic null model.
- Troubleshooting Step: Validate your permutation code. Ensure shuffling is performed correctly (e.g., within strata for a stratified design). Calculate a more precise p-value using the formula (R + 1) / (N + 1), where R is the number of permuted statistics >= observed, and N is the total permutations. Report as p < 0.0001 (for N=10,000).
- Thesis Relevance: An implausibly low p-value can mask overfitting if the permutation test does not accurately reflect the null hypothesis of "no association." An incorrect baseline inflates confidence in a potentially non-generalizable model.
Q2: My simulation results show extremely high variance in the null distribution of my performance metric (e.g., AUC). What does this mean for my analysis? A: High variance in your simulated null distribution suggests that the metric is unstable under random conditions given your dataset's characteristics (sample size, class imbalance, noise).
- Potential Issue: The performance metric may be unsuitable for your small or imbalanced dataset, as random chance can produce widely varying scores. This makes it difficult to distinguish a truly good model from a lucky one.
- Troubleshooting Step: Document the variance (e.g., standard deviation) of the null distribution in your results. Consider using a more robust metric or applying correction techniques. Increase your sample size if possible. The wide null baseline directly highlights the risk of overfitting to spurious patterns in limited data.
- Thesis Relevance: A wide null distribution sets a more challenging hurdle for claiming model efficacy. It quantifies the "noise floor," emphasizing that only performance significantly above this variable baseline should be considered non-random.
Q3: How do I choose between a simulation-based and a permutation-based approach to establish a random baseline? A: The choice depends on the specific null hypothesis you need to test.
- Permutation Testing is best when you want to test the null hypothesis of no association between the observed data labels and the observed data points. It works by randomly shuffling the outcome labels (or conditions) relative to the features, preserving the inherent data structure.
- Simulation is best when you need to generate entirely new data under a specific null model (e.g., data drawn from a Gaussian distribution with mean zero, or data with a specified correlation structure but no true class difference). It tests if your observed data could arise from a more abstract, theorized random process.
- Troubleshooting Guide: See the workflow diagram below for decision logic.
- Thesis Relevance: Selecting the appropriate baseline method is crucial. Using simulation when permutation is needed (or vice versa) can create an inaccurate or irrelevant baseline, leading to incorrect conclusions about model overfitting.
Q4: In permutation tests for classifier evaluation, should I permute labels before or after data splitting (train/test)? A: Permutation must be performed anew for each iteration, mimicking the entire model training and evaluation process under the null.
- Potential Issue: Permuting labels only once and then performing cross-validation leads to data leakage, as the permutation is fixed before splitting, creating an artificially optimistic or pessimistic null distribution.
- Correct Protocol: For a valid test, the entire procedure (label permutation -> data splitting -> model training on the training fold -> evaluation on the test fold) must be repeated for each permutation. This ensures the null hypothesis is consistently that the relationship between features and labels is random for that specific permutation.
- Thesis Relevance: Incorrect permutation during cross-validation is a common source of over-optimism (anti-conservative bias), undermining the very goal of detecting overfitting. It provides a falsely favorable baseline, making a tuned but overfit model appear significant.
Experimental Protocols
Protocol 1: Permutation Test for a Machine Learning Classifier's AUC Objective: To determine if the Area Under the ROC Curve (AUC) of a trained classifier is significantly better than chance. Method:
- Train your classifier on the original dataset (with true labels) and calculate the observed performance metric (e.g., AUC) using a held-out test set or via cross-validation. Record this value as
AUC_obs. - Define the number of permutations (e.g., N = 1000 or 10,000).
- For i in 1 to N:
a. Randomly shuffle (permute) the outcome labels (Y) across the entire dataset, breaking any true association between features (X) and labels.
b. Using the same data splitting scheme (e.g., the same CV folds) as in Step 1, retrain the classifier on the training portion of the permuted data.
c. Evaluate the classifier on the test portion of the permuted data, calculating the permuted AUC,
AUC_perm[i]. - Construct the null distribution from the list of all
AUC_permvalues. - Calculate the p-value as: (number of times
AUC_perm[i]>=AUC_obs) / N. For a more accurate, bias-corrected p-value, use: (count + 1) / (N + 1). - If p-value < your significance threshold (e.g., 0.05), reject the null hypothesis that the model's performance is consistent with random labeling.
Protocol 2: Simulation to Establish a Baseline for Correlation Analysis Objective: To assess whether an observed correlation coefficient (r) between two variables is stronger than expected by random noise in a small sample. Method:
- Calculate the observed correlation
r_obsfrom your experimental data of sample sizen. - Define your null model. A common choice is two independent, normally distributed variables. Define the number of simulations (e.g., S = 10,000).
- For i in 1 to S:
a. Generate two random vectors,
X_simandY_sim, each of lengthn, from a standard normal distributionN(0,1). This ensures no true correlation exists by construction. b. Calculate the correlation coefficientr_sim[i]betweenX_simandY_sim. - Construct the null distribution from the
r_simvalues. - Calculate the two-tailed p-value as: (number of times
abs(r_sim[i])>=abs(r_obs)) * 2 / S. Apply the (+1) correction as needed. - Compare the distribution of
r_simto yourr_obs. The simulation provides the range of correlation values achievable by random chance alone for your givenn.
Data Presentation
Table 1: Example Null Distribution Summary from a Permutation Test (Classifier AUC)
| Metric | Observed Value | Mean of Null (Permuted) | Std. Dev. of Null | 95% Percentile of Null | P-value (N=10,000) |
|---|---|---|---|---|---|
| AUC | 0.85 | 0.51 | 0.07 | 0.63 | < 0.0001 |
| Balanced Accuracy | 0.80 | 0.50 | 0.08 | 0.65 | 0.0003 |
Table 2: Impact of Sample Size on Simulated Null Distribution for Correlation
| Sample Size (n) | Mean Simulated |r| | Std. Dev. of Simulated |r| | 97.5% Percentile (Threshold) |
|---|---|---|---|
| 10 | 0.33 | 0.24 | 0.76 |
| 30 | 0.18 | 0.14 | 0.47 |
| 100 | 0.10 | 0.07 | 0.25 |
Visualizations
Title: Permutation Test Workflow for Model Evaluation
Title: Decision Guide: Simulation vs. Permutation Test
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Simulation & Permutation Studies
| Item/Category | Function & Relevance to Baseline Establishment |
|---|---|
| Statistical Software (R/Python) | Core environment for scripting permutation loops (sample() in R, np.random.permutation in Python) and simulations (rnorm, np.random.randn). Enables reproducibility. |
| High-Performance Computing (HPC) Cluster or Cloud Compute | Permutation tests (N>10,000) and complex simulations are computationally intensive. Parallel computing frameworks (e.g., foreach, multiprocessing) are essential reagents for timely analysis. |
| Random Number Generator (RNG) | The quality of the RNG (e.g., Mersenne Twister) and proper seeding (setting a seed for reproducibility) are critical. A flawed RNG invalidates the random baseline. |
| Stratification Variables | In complex designs, these are "reagents" to control for confounding (e.g., batch, patient cohort). Permutation must often be performed within strata to create a valid null dataset that preserves these structures. |
| Null Model Specification | The formal mathematical definition of the null hypothesis (e.g., "labels are exchangeable," "data is i.i.d. from distribution F"). This is the conceptual blueprint for generating the random baseline. |
| Performance Metric Library | A collection of evaluation functions (AUC, accuracy, precision, R², etc.) to calculate on both observed and null data, allowing comparison across different axes of model performance. |
Troubleshooting Guides & FAQs
Q1: My model performs exceptionally well on training data but fails on validation data. How can I quickly determine if this is overfitting? A: This is a classic sign of overfitting. Perform a rapid diagnostic by comparing key performance metrics between training and validation sets. A large gap (>15-20%) typically indicates overfitting. Implement a learning curve analysis by plotting training and validation scores against sample size; if the validation score plateaus well below the training score, your model is overfit.
Q2: After simplifying my neural network architecture to combat overfitting, the model now performs poorly on both sets. What step did I miss? A: You may have oversimplified, leading to underfitting. The goal is to find the "Goldilocks zone" of model complexity. Systematically increase capacity (e.g., add back layers/units) while employing strong regularization (e.g., Dropout, L2 regularization) after each increment. Monitor the performance gap. Use the following table to diagnose:
| Model Behavior | Training Accuracy | Validation Accuracy | Likely Cause | Corrective Action |
|---|---|---|---|---|
| Optimal Fit | High | High (gap < 5%) | Appropriate complexity | Maintain course. |
| Overfitting | Very High | Significantly Lower | Excessive complexity | Simplify model, increase regularization, gather more data. |
| Underfitting | Low | Equally Low | Insufficient complexity | Increase model capacity, reduce regularization, engineer better features. |
Q3: I have limited biological samples for drug response prediction. Which data augmentation strategies are most valid for tabular experimental data? A: For non-image biological data, use scientifically plausible augmentations:
- Noise Injection: Add small Gaussian noise to continuous measurements (e.g., gene expression, IC50 values) to simulate experimental variance.
- Mixup: Create synthetic samples via linear interpolations between real samples and their labels, which acts as a strong regularizer.
- SMOTE (Synthetic Minority Over-sampling Technique): Generates synthetic samples for underrepresented classes by interpolating between nearest neighbors in feature space.
- Domain-Specific Transformations: If using temporal data (e.g., cell growth curves), apply random time warping or scaling within biologically feasible bounds.
Protocol: Validating Augmentation for Molecular Data
- Split: Reserve a pristine, non-augmented test set.
- Augment: Apply chosen techniques (e.g., noise injection at 5% std dev) only to the training set.
- Train & Validate: Train model on augmented training data. Evaluate on the non-augmented validation set.
- Benchmark: Compare performance against the model trained without augmentation on the pristine test set. Valid augmentation should improve test accuracy.
Q4: Increasing sample size is often prohibitive in early drug discovery. What is a statistically sound minimal sample increase target? A: Conduct a power analysis or use learning curves. A rule-of-thumb target is to increase your sample size until the validation error confidence interval overlaps with the training error curve. If doubling the sample size is impossible, aim for a 20-30% increase combined with aggressive regularization, as shown in the simulated impact below:
| Sample Size | Avg. Training Error | Avg. Validation Error | Error Gap | Recommended Action |
|---|---|---|---|---|
| N = 50 | 0.02 | 0.25 | 0.23 | Severe overfitting. Increase N + simplify model. |
| N = 100 | 0.05 | 0.18 | 0.13 | Moderate overfitting. Apply regularization. |
| N = 200 | 0.08 | 0.12 | 0.04 | Good balance. Maintain. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Overfitting Mitigation |
|---|---|
| Dropout Regularization Reagents | Chemical analogs in computational models; randomly "drops out" neurons during training to prevent co-adaptation and over-reliance on specific features. |
| L1/L2 Regularization Optimizers | Algorithms (e.g., AdamW) that penalize model complexity by adding a term to the loss function proportional to the absolute (L1) or squared (L2) magnitude of weights. |
| Data Augmentation Libraries | Software tools (e.g., Imgaug for images, SMOTE-variants for tabular data) that generate synthetic, label-preserving training samples to effectively increase dataset size and diversity. |
| Cross-Validation Frameworks | Tools (e.g., scikit-learn KFold, StratifiedKFold) that partition data into multiple train/validation splits to ensure robust performance estimation and hyperparameter tuning. |
| Early Stopping Callbacks | Monitoring functions that halt training when validation performance plateaus or degrades, preventing the model from learning noise in the training data. |
Visualizing the Corrective Measures Workflow
Title: Decision Workflow for Addressing Model Overfitting
Data Augmentation Validation Protocol
Title: Valid Data Augmentation and Testing Workflow
Technical Support Center: Biomarker Signature Validation
FAQ & Troubleshooting Guide
Q1: Our initial biomarker panel performed perfectly on our training cohort but failed completely in the validation cohort. What is the most likely issue? A: This is a classic symptom of overfitting. The signature has learned noise or cohort-specific idiosyncrasies rather than a generalizable biological signal. Immediate troubleshooting steps include:
- Re-analyze Feature Selection: Review the feature selection method. Univariate filtering without cross-validation or correction for multiple testing often leads to overfitting.
- Check Cohort Balance: Ensure training and validation cohorts are matched for critical covariates (e.g., age, batch, clinical stage).
- Simplify the Model: Apply regularization (LASSO, Ridge) or reduce the number of features to the minimum required.
Q2: How can I distinguish a genuinely predictive biomarker from one selected by chance during high-dimensional screening? A: Implement rigorous statistical guards during discovery:
- Use Hold-Out or Cross-Validation: Perform all steps, including feature selection, within each cross-validation fold. Never use the full dataset to select features before CV.
- Apply Permutation Testing: Repeat your entire discovery pipeline on data where the outcome labels are randomly permuted. The performance on permuted data establishes a null distribution. Your real signature's performance must significantly exceed this.
Q3: What are the essential validation steps after identifying a potential signature from public omics data (e.g., TCGA)? A: A robust validation protocol requires technical, biological, and methodological layers:
- Technical/Platform Validation: Validate using an orthogonal measurement technique (e.g., move from RNA-seq to qPCR or immunohistochemistry).
- Independent Cohort Validation: Test in a completely independent cohort from a different institution or study.
- Prospective Validation: If possible, design a prospective study to assess the signature's predictive power in a real-world clinical workflow.
Q4: Our validation results are directionally consistent but statistically weaker. Can the signature still be salvaged? A: Yes. This common result suggests a core signal exists but was inflated. Salvage strategies include:
- Meta-Analysis: Systematically gather all available public and private datasets for the disease context and perform a meta-analysis to obtain a robust effect size estimate.
- Signature Refinement: Re-train the model on a larger, aggregated dataset using stricter regularization, or refine the panel to the most robust 2-3 biomarkers.
Data Presentation: Comparison of Signature Performance
Table 1: Performance Metrics of Hypothetical Biomarker Signature 'OncoSig-12' Across Analysis Phases
| Analysis Phase | Cohort (N) | Number of Features | AUC (95% CI) | Accuracy | Sensitivity | Specificity | Notes |
|---|---|---|---|---|---|---|---|
| Initial Discovery | TCGA Training (n=300) | 12 | 0.98 (0.96-1.00) | 0.95 | 0.96 | 0.94 | Severe overfitting evident |
| Internal CV | TCGA Full (n=300) | 12 | 0.71 (0.65-0.77) | 0.68 | 0.75 | 0.62 | Nested CV reveals true performance |
| Re-analysis (LASSO) | TCGA Training (n=300) | 4 | 0.87 (0.82-0.92) | 0.82 | 0.85 | 0.80 | Regularization applied |
| Independent Validation | GEO: GSE12345 (n=150) | 4 | 0.81 (0.74-0.88) | 0.78 | 0.80 | 0.76 | Generalizable signal confirmed |
| Orthogonal Validation | In-house IHC Cohort (n=80) | 4 | 0.79 (0.68-0.90) | 0.76 | 0.78 | 0.74 | Technical validation successful |
Experimental Protocols
Protocol 1: Nested Cross-Validation for Biomarker Discovery Objective: To obtain an unbiased performance estimate for a signature developed from high-dimensional data.
- Outer Loop: Split data into k-folds (e.g., k=5).
- Inner Loop: For each training set in the outer loop, perform another k-fold CV to optimize model parameters (including feature selection).
- Feature Selection: Select features using only the inner loop training folds. Common methods: LASSO regression with tuning via inner CV.
- Model Training: Train a final model (e.g., logistic regression) with the selected features on the outer loop's training fold.
- Testing: Apply the trained model to the held-out outer test fold. Repeat for all outer folds.
- Performance Aggregation: Calculate final metrics (AUC, accuracy) by aggregating predictions from all held-out test folds.
Protocol 2: Orthogonal Technical Validation via qPCR Objective: To validate an RNA-seq-derived gene expression signature using quantitative PCR.
- Sample: Use the same RNA samples from the discovery cohort or an independent set.
- cDNA Synthesis: Reverse transcribe 1µg of total RNA using a high-capacity cDNA kit with random primers.
- Assay Design: Design TaqMan assays or SYBR Green primers for the target biomarkers and 2-3 stable reference genes (e.g., GAPDH, ACTB, HPRT1).
- qPCR Run: Perform reactions in triplicate on a 384-well plate. Include no-template controls.
- Analysis: Calculate ∆Ct values (Ct[target] - Ct[reference geometric mean]). Use the ∆∆Ct method to compare between case and control groups. Perform statistical testing (t-test/Mann-Whitney) on ∆Ct or ∆∆Ct values.
Mandatory Visualizations
Title: Workflow for Salvaging an Overfit Biomarker Signature
Title: Nested Cross-Validation Pipeline to Prevent Overfitting
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents & Kits for Biomarker Validation Studies
| Item | Function/Benefit | Example Supplier/Kit |
|---|---|---|
| High-Capacity cDNA Reverse Transcription Kit | Converts RNA to stable cDNA with high efficiency and reproducibility, crucial for downstream qPCR. | Thermo Fisher Scientific (Cat #4368814) |
| TaqMan Gene Expression Assays | Predesigned, highly specific probe-based assays for quantitative real-time PCR validation. | Thermo Fisher Scientific |
| SYBR Green Master Mix | Cost-effective, flexible dye-based chemistry for qPCR; requires rigorous primer optimization. | Bio-Rad (SsoAdvanced) |
| NanoString nCounter Panels | Enables multiplexed digital quantification of up to 800 targets without amplification, ideal for orthogonal validation. | NanoString Technologies |
| Multiplex Immunohistochemistry Kit | Allows simultaneous detection of 4+ protein biomarkers on a single FFPE tissue section for spatial validation. | Akoya Biosciences (OPAL) |
| RNeasy FFPE Kit | Extracts high-quality RNA from formalin-fixed, paraffin-embedded (FFPE) tissues for retrospective cohort analysis. | Qiagen |
| LASSO Regression Software Package | Performs regularized regression to select the most predictive features and avoid overfitting. | R package 'glmnet' |
Proving Robustness: Validation Frameworks and Comparative Best Practices
In the context of method development and evaluation research, particularly in drug development, validation is a critical guardrail against overfitting. Overfitting occurs when a model describes random error or noise instead of the underlying relationship, leading to excellent performance on training data but poor generalizability. Internal and external validation are two fundamental approaches used to assess and ensure the robustness and predictive ability of analytical methods, algorithms, and statistical models.
Definitions
Internal Validation: A set of procedures used to evaluate a model's performance using resampling techniques (e.g., cross-validation, bootstrap) on the original dataset. It provides an estimate of model performance from the data used in development.
External Validation: The process of evaluating a model's performance on entirely new, independent data not used in any part of the model development or training process. This is the gold standard for assessing generalizability.
Purposes
Purpose of Internal Validation:
- To provide an optimistic but reasonable estimate of model performance during development.
- To guide model selection and hyperparameter tuning.
- To mitigate overfitting by simulating performance on unseen data within the available dataset.
- To be used when acquiring a fully independent external dataset is impractical or costly.
Purpose of External Validation:
- To provide an unbiased, real-world assessment of a model's predictive performance and generalizability.
- To confirm that the model works in different populations, settings, or with different measurement instruments.
- To fulfill regulatory requirements for robust method qualification in drug development.
- To provide the strongest evidence that a model is not overfit to its development data.
Implementation: A Technical Support Guide
This section addresses common issues researchers face when implementing validation strategies.
Troubleshooting Guides & FAQs
Q1: Our model achieves >90% accuracy in 10-fold cross-validation but performs poorly (<60%) when deployed. What went wrong?
- Likely Cause: Fundamental flaws in the internal validation setup leading to data leakage, or a significant covariate shift between your development and deployment data.
- Troubleshooting Steps:
- Audit Data Splitting: Ensure your cross-validation splits respect the independence of samples. For time-series or patient data, splits must be chronological or by patient ID to prevent leakage.
- Check Preprocessing: All preprocessing (imputation, scaling, normalization) must be fit only on the training fold and applied to the validation fold. Fitting on the entire dataset before splitting leaks global information.
- Perform a Covariate Shift Analysis: Statistically compare the distributions of key features between your development and deployment datasets. Significant differences explain the performance drop.
Q2: We lack the resources to collect a large, independent external validation cohort. What are our options?
- Solution: Consider robust internal validation or a hybrid approach.
- Use Nested Cross-Validation: This is the most rigorous internal method. An outer loop estimates unbiased performance, while an inner loop handles model tuning. It prevents optimistic bias from using the same data for tuning and performance estimation.
- Leverage Public Repositories: For common assay types (e.g., gene expression, proteomics), validate your model on curated public datasets from repositories like GEO, PRIDE, or TCGA.
- Collaborative Validation: Propose a validation swap with a peer laboratory using a comparable but independent dataset.
Q3: How do we decide the proportion of data for training, internal validation (tuning), and external validation (hold-out) sets?
- Guidelines: There is no universal rule, as it depends on total sample size and model complexity.
- Decision Table:
| Total Sample Size | Recommended Strategy | Rationale |
|---|---|---|
| Small (n < 100) | Repeated K-fold (K=5 or 10) or Bootstrapping. Avoid a separate hold-out test set. | Maximizes use of limited data for training; a hold-out set would be too small for reliable performance estimation. |
| Medium (100 < n < 1000) | Train/Validation/Hold-out split (e.g., 70%/15%/15%) or Nested Cross-Validation. | Allows for a reasonably sized hold-out test while maintaining adequate training data. |
| Large (n > 1000) | Train/Validation/Hold-out split (e.g., 80%/10%/10%). | Ensures ample, independent data for both tuning and final, high-confidence evaluation. |
Q4: What are the key performance metrics to report for internal vs. external validation?
- Answer: Report the same core metrics for both to allow direct comparison. The discrepancy between them is a key indicator of overfitting.
- Summary Table for a Binary Classifier:
| Metric | Formula/Purpose | Internal Validation Report | External Validation Report |
|---|---|---|---|
| Accuracy | (TP+TN)/(TP+TN+FP+FN) | Report mean (± SD) across folds. | Report single value on hold-out set. |
| Precision | TP/(TP+FP) | Report mean (± SD) across folds. | Report single value on hold-out set. |
| Recall (Sensitivity) | TP/(TP+FN) | Report mean (± SD) across folds. | Report single value on hold-out set. |
| AUC-ROC | Area under ROC curve | Report mean (± SD) across folds. | Report single value on hold-out set. |
| Calibration | Agreement of predictions with observed outcomes | Check on pooled out-of-fold predictions. | Mandatory: Report via calibration plot/statistics. |
Experimental Protocol: Nested Cross-Validation for Robust Internal Validation
Objective: To obtain an unbiased estimate of model performance while performing hyperparameter tuning, minimizing the risk of overfitting and data leakage.
Materials: Labeled dataset, computational environment (e.g., Python/R).
Procedure:
- Define Outer Loop (Performance Estimation): Split the entire dataset into K outer folds (e.g., K=5 or 10). For each outer fold i: a. Set aside fold i as the outer test set. b. The remaining K-1 folds constitute the outer training set.
- Define Inner Loop (Model Tuning): On the outer training set, perform a second, independent L-fold cross-validation (e.g., L=5). a. This inner loop is used to train and evaluate models with different hyperparameter combinations. b. Select the hyperparameter set that yields the best average performance across the L inner validation folds.
- Train Final Inner Model: Train a new model on the entire outer training set using the optimal hyperparameters identified in Step 2.
- Evaluate: Apply this final model to the held-out outer test set (fold i) from Step 1 to obtain a performance estimate P_i.
- Repeat: Iterate Steps 1-4 for all K outer folds.
- Final Performance Estimate: Calculate the mean and standard deviation of the K performance estimates (P_1...P_K). This is the unbiased internal validation performance.
Visualization: Validation Workflow & Decision Pathway
Title: Decision Pathway for Selecting a Validation Strategy
The Scientist's Toolkit: Research Reagent Solutions for Validation Studies
| Reagent / Material | Function in Validation Context |
|---|---|
| Certified Reference Material (CRM) | Provides a ground truth with known, traceable properties. Essential for external validation of analytical method accuracy across labs. |
| Independent Cohort Biospecimens | Fresh, biologically relevant samples collected under a distinct protocol. The cornerstone of external validation to test generalizability. |
| Synthetic Control Spikes | Artificially introduced molecules (e.g., SIS peptides in proteomics) used to monitor and correct for technical variation during both internal and external validation runs. |
| Benchmarking Datasets (Public) | Curated, public datasets (e.g., from NIH repositories) serving as a cost-effective external validation standard for computational models. |
| Data Partitioning Software/Libraries | Tools (e.g., scikit-learn train_test_split, GroupKFold) that ensure correct, reproducible, and leak-free splitting of data for robust validation setups. |
| Calibration Plot Analysis Tools | Software (e.g., val.prob in R, calibration_curve in sklearn) to assess prediction reliability, a mandatory check in external validation reports. |
Designing a Robust External Validation Study with Independent Cohorts
Technical Support Center: Troubleshooting & FAQs
Frequently Asked Questions
Q1: Our model performed excellently in internal validation but failed completely in the external cohort. What is the most likely cause? A1: This is a classic symptom of overfitting. The model likely learned noise, batch effects, or site-specific artifacts from your development data rather than the true biological signal. Ensure your internal validation used truly independent data (not just random splits) and consider implementing stricter regularization during development.
Q2: How many independent cohorts are sufficient for a robust external validation? A2: While more is always better, a minimum of two independent, high-quality cohorts is considered essential. One cohort acts as the primary validation set, while the second confirms generalizability. The key is not just the number, but their diversity in terms of demographics, sample collection protocols, and sequencing/platform batches.
Q3: What are the key differences between a test set, a validation set, and an external cohort? A3:
| Set Type | Purpose | Data Source | Timing of Access |
|---|---|---|---|
| Training Set | Model development & parameter fitting. | Initial, single-source cohort. | First. |
| (Internal) Validation Set | Tuning hyperparameters & selecting best model iteration. | Held-out portion of the initial cohort. | During development. |
| Test Set | Final, unbiased estimate of performance pre-deployment. | Held-out portion of the initial cohort, untouched until the very end. | After model is fully locked. |
| External Validation Cohort(s) | Assessing generalizability and real-world performance. | Entirely independent cohort(s) from different sites/populations. | After model is fully locked. |
Q4: How should we handle batch effect correction between the development and external cohorts?
A4: Apply any correction algorithm (e.g., ComBat, limma's removeBatchEffect) only to the development data, using parameters learned from it. Then, apply that same transformation to the external data. Never correct all data together, as this artificially removes inter-cohort differences and leads to over-optimistic performance (data leakage).
Q5: What performance metrics are most meaningful for external validation? A5: Prioritize metrics that are robust to class imbalance and clinically interpretable. Report a suite of metrics, not just one.
| Metric | Best For | Caution |
|---|---|---|
| Area Under the ROC Curve (AUC) | Overall discriminative ability. | Can be optimistic for imbalanced data. |
| Average Precision (AUPRC) | Imbalanced datasets. | Less intuitive clinically. |
| Calibration Slope & Intercept | Assessing prediction reliability. | Critical for risk models. |
| Sensitivity/Specificity at a clinically relevant threshold | Clinical utility. | Threshold must be pre-specified. |
Experimental Protocols for Key Validation Steps
Protocol 1: Pre-Validation Cohort Auditing
- Objective: Assess the suitability of an independent cohort for validation.
- Materials: Metadata for both development and proposed external cohorts.
- Procedure: a. Compare population demographics (age, sex, ethnicity) and clinical characteristics. b. Audit sample procurement, processing protocols, and storage conditions for major discrepancies. c. For genomic/proteomic data, perform Principal Component Analysis (PCA) to visualize batch and platform effects. d. Document all differences in a standardized table. Decide if differences are strengths (testing generalizability) or fatal flaws (incomparable populations).
- Deliverable: A cohort comparison report justifying the use of the external dataset.
Protocol 2: Locking the Analysis Plan
- Objective: Prevent analytical flexibility ("researcher degrees of freedom") from inflating performance.
- Procedure:
a. Before applying the model to the external cohort, finalize and document in a protocol:
- The exact, final model (including coefficients/weights).
- The pre-processing steps (imputation, normalization, feature scaling) with all parameters.
- The primary and secondary performance metrics.
- The statistical methods for calculating confidence intervals. b. Register this protocol on a public repository (e.g., OSF, GitHub) for time-stamping.
- Deliverable: A time-stamped, immutable analysis plan.
Protocol 3: Executing the Blinded Validation
- Objective: Obtain an unbiased performance estimate.
- Procedure: a. The external cohort data (features) are prepared by a biostatistician following the locked pre-processing plan. b. The locked model is applied to generate predictions. c. These predictions are sent to a separate analyst who holds the ground truth labels. This analyst calculates all pre-specified performance metrics without any further model tweaking. d. Results are compiled and compared against the development performance.
Visualizations
External Validation Study Workflow
Overfitting Causes and External Validation Solution
The Scientist's Toolkit: Research Reagent Solutions
| Item / Solution | Function in External Validation | Example / Note |
|---|---|---|
| Public Genomic/Clinical Repositories | Source of independent validation cohorts. | dbGaP, GEO, EGA, TCGA, UK Biobank. Ensure appropriate data use agreements. |
| Batch Effect Correction Algorithms | Standardize data across cohorts without leaking information. | ComBat (sva package), limma's removeBatchEffect. Use with caution. |
| Containerization Software | Ensures computational reproducibility of the locked model. | Docker, Singularity. Package the exact software environment. |
| Version Control Systems | Tracks every change to the analysis code, creating an audit trail. | Git with platforms like GitHub or GitLab. |
| Electronic Lab Notebook (ELN) | Documents the validation protocol, cohort metadata, and decisions. | Platforms like LabArchives, Benchling. Critical for auditability. |
| Statistical Analysis Platforms | For pre-specified, reproducible statistical evaluation. | R Markdown, Jupyter Notebooks. Weave code, results, and commentary. |
| Biomarker/Sample Quality Assay Kits | To confirm sample/assay quality in the external cohort matches expectations. | RNA Integrity Number (RIN) assays, immunohistochemistry controls. |
Technical Support Center: Troubleshooting Guides & FAQs
FAQ 1: Data Integration & Preprocessing
Q: I have merged RNA-Seq data from TCGA and GEO, but my batch effect correction (using ComBat) is not working. The PCA plot still shows strong separation by dataset origin. What are the critical checks?
A: First, verify that your data is properly normalized (e.g., TPM, FPKM) and log2-transformed before applying ComBat. Ensure your model matrix (mod) correctly specifies the biological variable of interest (e.g., tumor vs. normal) and does not accidentally include the batch variable. Crucially, check for zero-variance or near-zero-variance genes across batches; these should be removed as they can destabilize the correction. Confirm you are using an appropriate reference batch if your version of ComBat requires it.
Q: When downloading TCGA data via the GDC API, my clinical and genomic feature tables do not align. Key samples are missing. How do I resolve this? A: This is a common issue due to differing data availability. Follow this protocol:
- Always use the
gdc.casesendpoint to get the master list of cases (patients). - For each data type (e.g., clinical, mRNA expression), query the
gdc.filesendpoint, filtering bycases.submitter_idanddata_type. - Use the
related_casesendpoint to map file IDs to case IDs reliably. - Perform an inner join on
case_idacross all your data tables. This ensures you only work with the subset of samples present in all required data modalities, preventing silent sample loss during analysis.
FAQ 2: Synthetic Data Generation & Validation Q: My synthetic data generated using a GAN fails to capture the covariance structure of the real TCGA data. What parameters should I audit? A: This indicates a failure in model training or architecture.
- Architecture Check: Increase the depth or width of the generator and discriminator networks. Consider using a Wasserstein GAN (WGAN) with Gradient Penalty (GP) for improved stability.
- Training Audit: Ensure you are training for a sufficient number of epochs. Monitor the loss functions—they should oscillate, not diverge. Validate that the synthetic data passes statistical tests (e.g., KS test for marginal distributions, comparison of principal components) at multiple checkpoints during training.
- Input Noise: Verify the dimensionality and distribution (e.g., Gaussian) of the input noise vector
z; it should have sufficient complexity to model the output space.
Q: How do I rigorously benchmark a new classification method to prove it is robust and not overfit to a specific dataset? A: Implement a tiered benchmarking strategy as per the thesis on mitigating overfitting:
Table 1: Tiered Benchmarking Protocol for Robustness Validation
| Tier | Data Type | Purpose | Key Validation Metric | Success Criterion |
|---|---|---|---|---|
| T1 | Synthetic Data (Controlled) | Test method logic under ideal, known conditions. | Precision, Recall, F1-Score | Performance > 0.95 on all metrics. |
| T2 | Curated Public Data (e.g., TCGA, split by cohort) | Assess performance on real but potentially confounded data. | AUC-ROC, Balanced Accuracy | Performance consistent across 5 random 80/20 train/test splits (SD < 0.05). |
| T3 | Independent Hold-Out Set (e.g., a separate GEO dataset) | Evaluate generalizability to new populations/labs. | AUC-ROC, Cohen's Kappa | Performance drop from T2 < 0.10. |
| T4 | "Noisy" or Perturbed Data | Stress-test robustness to missing values and noise. | Degradation of AUC-ROC | Performance drop from T2 < 0.15 after adding 10% missingness. |
Experimental Protocol for Tiered Benchmarking:
- Synthetic Data Generation: Use a tool like
scikit-learn'smake_classificationor a fitted Gaussian Copula to generate data with predefined cluster structures and feature correlations. - Data Splitting: For T2, strictly separate a hold-out test set (20%) before any exploratory analysis or feature selection. Use the remaining 80% for cross-validation.
- Hyperparameter Tuning: Conduct tuning only on the T2 training fold(s) using nested cross-validation.
- Final Evaluation: Train the final model with optimal parameters on the entire T2 training set and evaluate once on the T2 test set and the completely independent T3 dataset.
- Perturbation Test (T4): Randomly introduce missing values (MCAR) or Gaussian noise into the T3 dataset features and re-evaluate.
FAQ 3: Pathway & Workflow Analysis Q: My pathway enrichment analysis yields different results when run on TCGA data vs. synthetic data simulating TCGA. Is this expected? A: Yes, but within limits. Synthetic data should approximate, not perfectly replicate, complex biological networks. Use the following workflow to diagnose discrepancies:
Diagram 1: Validating Pathway Enrichment Concordance
If the process in Diagram 1 fails, inspect the synthetic data's gene-gene correlation matrix versus the real data's. Large discrepancies here will cause divergent pathway results.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Tools for Benchmarking & Analysis
| Item / Resource | Function / Purpose | Key Consideration for Overfitting Mitigation |
|---|---|---|
| cBioPortal | Interactive exploration of multidimensional cancer genomics data (TCGA, etc.). | Use for hypothesis generation only. Any observation must be validated in a formal, held-out test set. |
| GDCRNATools / TCGAbiolinks | R packages for streamlined TCGA data download, integration, and analysis. | Always use the data.type="normalized" parameter for comparable counts. Split data by project_id for cohort-based validation. |
scikit-learn Pipeline |
Python tool to chain preprocessing and model steps. | Encapsulates all transformations, preventing information leak from test data into training process during cross-validation. |
| SynthCity | Python library for generating synthetic tabular data (GANs, VAEs, CTGAN). | Use its Metrics.evaluate module to quantitatively assess how well synthetic data preserves real data's statistical properties and predictive utility. |
| MLxtend | Python library providing RandomHoldoutSplit and PredefinedHoldoutSplit. |
Crucial for creating clean, reproducible train/test splits, especially when using multiple sourced datasets (TCGA + GEO). |
| UCSC Xena | Public hub for hosting and visualizing functional genomics datasets. | Its "cohort selection" feature is ideal for quickly creating independent external validation sets from non-TCGA studies. |
Diagram 2: Core Workflow to Prevent Data Leakage
Technical Support Center: Troubleshooting & FAQs
Q1: My model validation performance drops significantly compared to training. I suspect overfitting. How can reporting standards like TRIPOD help me diagnose and rectify this?
A: TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis) mandates a complete reporting of model development and validation. First, ensure your report includes Table 1 (Participant Characteristics) for both development and validation cohorts. A mismatch here indicates a validation cohort that is not representative, leading to performance drop. Second, check TRIPOD Item 10b: Did you report the full prediction model, including all coefficients? If not, you may have inadvertently omitted a key variable, making the model unstable. The protocol is to: 1) Re-examine your development data for data leakage. 2) Apply stronger regularization (e.g., LASSO) during model development as per your pre-specified analysis plan (TRIPOD Item 10a). 3) Use the TRIPOD checklist to audit your own report; missing items often point to methodological flaws.
Q2: My microarray experiment is irreproducible. How can MIAME guidelines help me troubleshoot?
A: MIAME (Minimum Information About a Microarray Experiment) ensures all data necessary to interpret and replicate the experiment is available. Common issues and MIAME-based solutions:
- Problem: Inconsistent normalization leading to batch effects.
- MIAME Check: Did you fully document the "Normalization Controls" (MIAME Section 3)? Provide the exact protocol: Raw CEL files were normalized using the Robust Multi-array Average (RMA) algorithm in [Software, version]. The protocol must include the software, version, and all parameters used.
- Problem: Unable to match sample to data.
- MIAME Check: Document the "Sample Data" (MIAME Section 1) and "Array Data" (MIAME Section 4) meticulously. Create a sample-to-array matrix table. The protocol: For each sample, record unique identifier, experimental factor values (e.g., dose, time), and the exact array data file name it was hybridized to.
Q3: How do I choose the correct reporting guideline for my study to prevent overfitting in method development?
A: Selecting the appropriate guideline structures your study to avoid over-optimistic results. Use this decision table:
| Study Type | Primary Guideline | How it Mitigates Overfitting |
|---|---|---|
| Clinical Prediction Model Development/Validation | TRIPOD | Mandates separate reporting of development & validation cohorts, and complete model specification to prevent data dredging. |
| Microarray/Gene Expression | MIAME | Requires full array design and normalization details, preventing selective reporting of "best" normalized data. |
| Next-Generation Sequencing (NGS) | MINSEQE | Demands detailed sequencing and preprocessing steps, ensuring analytical choices are justified and reproducible. |
| Randomized Controlled Trials (RCTs) | CONSORT | Forces pre-registration of outcomes and analysis plan, reducing the risk of cherry-picking statistically significant results. |
| Systematic Reviews/Meta-Analyses | PRISMA | Requires a systematic search and selection process, minimizing selection bias in included studies. |
Q4: I developed a new assay method. What reporting standard should I follow to ensure my evaluation is robust against overfitting?
A: For novel biomedical assays, follow the STARD (Standards for Reporting Diagnostic Accuracy) guidelines. Crucially, it requires a flow diagram of participant inclusion. This prevents cherry-picking samples that perform well. The experimental protocol for evaluation must: 1) Pre-define the test's positive cutoff before evaluating against the reference standard (STARD Item 12). 2) Blind the assessors of the index test and reference standard to each other's results (STARD Item 11). 3) Report indeterminate/missing results (STARD Item 14). Document all samples attempted, not just the successful runs.
| Reporting Guideline | Field | Reported Improvement in Reproducibility (Study Year) | Median % Increase in Methodological Clarity Post-Adoption |
|---|---|---|---|
| TRIPOD | Prediction Modeling | 40% reduction in bias in validation estimates (2021) | 58% |
| MIAME | Genomics/Microarray | 3-fold increase in data reproducibility (2019) | 72% |
| CONSORT | Clinical Trials | 25% improvement in trial quality assessment (2020) | 64% |
| ARRIVE 2.0 | Animal Research | Significant increase in reporting of blinding, randomization (2022) | 49% |
| STARD | Diagnostic Test Accuracy | Improved accuracy of sensitivity/specificity estimates (2021) | 55% |
Experimental Protocol: Validating a Predictive Model under TRIPOD
Title: Protocol for Internal-External Cross-Validation to Combat Overfitting. Objective: To develop and validate a prognostic model while assessing its generalizability and mitigating overfitting.
- Cohort Segmentation: Partition your total dataset (N=) into K distinct geographical or temporal clusters (e.g., by study center or recruitment year).
- Iterative Training/Validation: For i = 1 to K: a. Set cluster i as the validation set. b. Pool the remaining K-1 clusters as the development set. c. Develop the model anew on the development set, including all steps (predictor selection, handling missing data, model fitting). d. Apply the developed model to validation cluster i and calculate performance (C-statistic, calibration slope).
- Performance Aggregation: Pool performance estimates from all K iterations to obtain an overall estimate of model performance and its heterogeneity.
- Final Model: Fit the final model using the entire dataset, reporting all coefficients as per TRIPOD Item 10b. The validation from Step 3 estimates its expected performance on new data.
Signaling Pathway: Reporting Standards Impact on Research Quality
Title: Guideline Use in Research Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item/Category | Function in Context of Transparent Reporting & Overfitting Mitigation |
|---|---|
| Versioned Code Repository (e.g., Git/GitHub) | Tracks all analytical code changes, ensuring the reported analysis matches the exact code used. Prevents "tuning" code to fit data. |
| Data & Metadata Standards (e.g., ISA-Tab) | Structures experimental metadata (sample, protocol, data file) in a machine-readable format, fulfilling MIAME/MINSEQE requirements. |
| Blinded Analysis Software/Protocol | Software or SOPs that enable blinding of analyst to experimental group during initial data processing, reducing subconscious bias. |
| Pre-registration Platform (e.g., OSF, ClinicalTrials.gov) | Documents hypotheses, primary outcomes, and analysis plan before experimentation, a core tenet of CONSORT and PRISMA. |
| Electronic Lab Notebook (ELN) with Audit Trail | Provides immutable, timed records of all experimental procedures and parameters, supporting detailed methodology sections. |
| Bioinformatics Pipelines (Versioned, e.g., Nextflow) | Encapsulates entire data analysis workflow (QC, normalization, modeling), ensuring computational reproducibility for peer review. |
Technical Support Center
Topic: Troubleshooting Validation Scheme Implementation
Troubleshooting Guides & FAQs
Q1: My model performs excellently during cross-validation but fails spectacularly on the final, held-out test set. What is the most likely cause and how can I diagnose it? A: This is a classic symptom of data leakage or an improper validation scheme. The model has been tuned or indirectly exposed to information from the test set during training/validation. To diagnose:
- Audit your preprocessing: Ensure all steps (imputation, scaling, normalization) are fit only on the training fold within each cross-validation (CV) split and then applied to the validation fold. Never fit on the entire dataset before splitting.
- Review your CV strategy: For time-series or structured data, random CV can create unrealistic leakage. Use schemes like
TimeSeriesSplit. For grouped data (multiple samples from same patient), useGroupKFold. - Implement nested cross-validation: Use an inner CV loop for hyperparameter tuning and an outer CV loop for performance estimation. This strictly separates tuning from evaluation.
Q2: How do I choose between k-fold cross-validation, leave-one-out, and a single hold-out set for my high-dimensional, small sample size (n=50, p=1000) omics dataset? A: For small-n-large-p scenarios:
- Avoid single hold-out: The test set would be too small to be statistically reliable.
- Use caution with Leave-One-Out (LOO): LOO has low bias but very high variance with small n, leading to unstable performance estimates. It also does not mimic a true train/test paradigm as effectively.
- Recommended: Repeated k-fold CV (e.g., 5-fold repeated 10 times): This provides a better balance of bias and variance. The repeated iterations allow you to assess the stability of your estimate. Ensure folds are stratified to preserve class distribution.
Q3: I'm implementing a new machine learning method and need to demonstrate it resists overfitting. What validation scheme is considered the "gold standard" for a definitive performance report in a publication? A: The current best practice is a three-tiered, locked-box approach:
- Training Set: For model development.
- Validation Set (or inner CV loop): For hyperparameter tuning and model selection.
- Final Test Set (or outer CV loop): Used ONLY ONCE for the final performance report. This set must be locked away from any development activity. The result from this single evaluation is what you report. Using an external validation cohort from a completely independent study is the most rigorous standard.
Q4: What are the key metrics to track, beyond mean accuracy, to evaluate the health and robustness of my validation scheme itself? A: Monitor the distribution and variance of your performance metrics across all folds/splits.
- Calculate: The mean, standard deviation, min, and max of your primary metric (e.g., AUC-ROC) across CV folds.
- Visualize: Use boxplots of fold-wise scores.
- Interpret: A large variance between folds suggests your model's performance is highly dependent on the data split, indicating potential instability, insufficient data, or data heterogeneity. A small variance increases confidence in your reported mean.
Experimental Protocols for Cited Key Experiments
Protocol 1: Nested Cross-Validation for Algorithm Comparison Objective: To fairly compare two classification algorithms (Algorithm A vs. Algorithm B) while minimizing overfitting from hyperparameter tuning.
- Define Outer Loop: Split the full dataset into k outer folds (e.g., k=5 or 10). Stratify by class label.
- Iterate Outer Loop: For each outer fold i: a. Designate fold i as the temporary test set. The remaining k-1 folds are the development set. b. On the development set, run an inner k-fold cross-validation (e.g., 5-fold). c. Within the inner loop, train and tune the hyperparameters for both Algorithm A and B independently. d. Select the best hyperparameter set for each algorithm based on the inner CV performance. e. Using these optimal hyperparameters, train each algorithm on the entire development set. f. Evaluate each trained model on the held-out outer fold i (temporary test set). Record the performance metric (e.g., AUC).
- Result: You will have k performance estimates for each algorithm. Perform a paired statistical test (e.g., paired t-test on the k AUC values) to determine if one algorithm is significantly better.
Protocol 2: External Validation with a Prospective Cohort Objective: To provide the highest level of evidence for a developed diagnostic model.
- Model Development Phase: Use all available retrospective data (Cohort R) for model training and hyperparameter tuning using nested CV as in Protocol 1. Finalize the model and its training protocol.
- Validation Phase: a. Prospectively define a new cohort (Cohort P) based on the intended use setting, with pre-defined inclusion/exclusion criteria and target sample size (power calculation). b. Apply the locked model: Process the new data from Cohort P using the exact, frozen preprocessing pipeline and model from Step 1. c. Single Evaluation: Calculate the model's performance metrics (sensitivity, specificity, etc.) on Cohort P once. d. Report these metrics with confidence intervals. This evaluation is unbiased by any development activity.
Data Presentation
Table 1: Comparison of Common Validation Schemes
| Scheme | Typical Use Case | Advantages | Disadvantages | Risk of Overfitting |
|---|---|---|---|---|
| Single Hold-Out | Large datasets (n > 10,000), initial quick checks. | Simple, fast, mimics real train/test. | High variance estimate, inefficient data use. | High if used repeatedly. |
| k-Fold CV | Standard for medium-sized datasets. | Reduces variance vs. hold-out, more efficient data use. | Computationally heavier; can be biased with structured data. | Moderate if not nested. |
| Stratified k-Fold | Classification with imbalanced classes. | Preserves class distribution in folds, less biased estimate. | Same as k-Fold CV. | Moderate if not nested. |
| Leave-One-Out (LOO) | Very small datasets (n < 100). | Low bias, deterministic. | High variance, computationally expensive for large n. | Low, but estimates unstable. |
| Repeated k-Fold | Small to medium datasets requiring stable estimate. | More stable/reliable estimate than single k-fold. | Increased computation. | Moderate if not nested. |
| Nested CV | Gold standard for tuning & evaluation on a single dataset. | Provides nearly unbiased performance estimate. | Computationally very expensive (O(k²)). | Very Low |
| External Validation | Final proof of generalizability before deployment. | Highest level of evidence, tests real-world performance. | Requires additional, independent data collection. | Lowest |
Table 2: Example Results from a Nested CV Study Comparing Classifiers
| Classifier | Mean AUC (Inner CV) | Std Dev (Inner CV) | Mean AUC (Outer Test) | Std Dev (Outer Test) | p-value (vs. Random Forest) |
|---|---|---|---|---|---|
| Random Forest | 0.92 | 0.03 | 0.87 | 0.05 | -- |
| SVM (RBF) | 0.94 | 0.02 | 0.85 | 0.07 | 0.12 |
| Logistic Regression | 0.88 | 0.04 | 0.79 | 0.06 | <0.01 |
| Simple Neural Net | 0.95 | 0.02 | 0.84 | 0.08 | 0.09 |
Note: The lower Outer Test AUC vs. Inner CV AUC for all models indicates proper separation of tuning and evaluation. The higher standard deviation in Outer Test reflects true performance variability.
Mandatory Visualizations
Title: Nested Cross-Validation Workflow
Title: Spectrum of Validation Scheme Rigor
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Robust Validation Experiments
| Item / Solution | Function in Validation | Example / Note |
|---|---|---|
scikit-learn (Python) |
Provides unified API for train_test_split, KFold, StratifiedKFold, GroupKFold, TimeSeriesSplit, and model fitting. Essential for implementing protocols. |
Use Pipeline to prevent data leakage. cross_val_score for simple CV. |
mlr3 or caret (R) |
Comprehensive machine learning frameworks in R that offer structured, reproducible interfaces for resampling, benchmarking, and hyperparameter tuning. | mlr3's nested resampling is explicitly designed for robust evaluation. |
random_state / Seed |
A numerical seed for pseudo-random number generators. Crucial for reproducibility of any random split (train/test, CV folds). | Always set this for any function that involves randomness (e.g., np.random.seed(42), random_state=42 in scikit-learn). |
pandas / DataFrame |
For careful, traceable data handling. Enables grouping, stratification, and ensures data integrity through splits. | Use .iloc for integer-location based splitting to avoid index misalignment issues. |
| Statistical Test Suite | To compare performance metrics between models or validation schemes statistically, not just by point estimates. | Use paired t-tests (for CV results), Delong's test (for AUC comparison), or McNemar's test (for accuracy). |
| Version Control (Git) | To track every change in code, model parameters, and data splitting logic. Allows exact replication of any reported result. | Commit code for each major experiment, including the final validation run. |
| Containerization (Docker) | Encapsulates the entire computational environment (OS, libraries, versions) to guarantee long-term reproducibility. | Ship a Docker image alongside your publication code. |
Technical Support Center: Troubleshooting Model Translation
FAQs & Troubleshooting Guides
Q1: Our validated prognostic model performs excellently on our internal cohort (AUC=0.94) but fails when tested on an external, multi-center dataset (AUC=0.62). What are the primary causes and corrective steps?
A: This classic sign of overfitting and lack of generalizability typically arises from:
- Data Leakage: Contamination between training and validation sets.
- Cohort-Specific Biases: Your internal cohort may not represent the broader patient population.
- Preprocessing Inconsistency: Differing normalization or imputation protocols between centers.
- Solution Protocol:
- Audit Data Splits: Re-check patient ID assignments to ensure no subjects are in both training and validation sets.
- Harmonize Preprocessing: Implement ComBat or similar batch-effect correction tools.
- Simplify the Model: Apply stronger regularization (L1/L2) or reduce feature number via recursive feature elimination.
- Validate with External Data Early: Use external data as a co-validation set from the method development phase.
Q2: During clinical assay development, our high-throughput sequencing biomarker signature loses predictive power when transferred to a clinically approved qPCR platform. How do we troubleshoot this?
A: This is a platform transfer issue. Follow this experimental protocol:
Experimental Protocol: Platform Transfer & Calibration
- Re-extract RNA: From the same original patient samples used in the discovery phase (if available).
- Parallel Testing: Run the RNA on both the original (NGS) and new (qPCR) platforms.
- Calibration Regression: For each biomarker in the signature, perform a linear regression between NGS (log-CPM) and qPCR (log-Ct) values. Establish a conversion formula.
- Re-threshold: Re-establish clinical cutoff points on the qPCR data using the outcome data, not the NGS-converted values.
- Lock Down the Protocol: Finalize the qPCR master mix, cycler, and sample prep SOPs before the next validation step.
Q3: We are preparing for a prospective clinical trial to validate our diagnostic model. What are the key statistical and regulatory checkpoints to avoid a failed trial due to methodological flaws?
A: Key checkpoints are summarized in the table below:
| Checkpoint Phase | Key Action | Quantitative Target | Common Pitfall |
|---|---|---|---|
| Pre-Trial: Assay Lock | Finalize and document the entire testing SOP. | All CVs < 15%. | Allowing "protocol drift" during the trial. |
| Pre-Trial: Sample Size | Power calculation based on clinical utility. | Power ≥ 90% for primary endpoint. | Powering only for accuracy (AUC), not clinical outcome. |
| Trial: Blinding | Ensure complete blinding of assay operators to clinical data. | 100% blinding audit success. | Unintentional unblinding via sample batch or date. |
| Analysis: Pre-Specification | File statistical analysis plan (SAP) before database lock. | All primary/secondary endpoints defined. | Performing unplanned subgroup analyses as primary findings. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Synthetic Spike-In Controls (e.g., ERCC RNA) | Added to patient samples pre-extraction to monitor technical variability and batch effects across experimental runs, crucial for longitudinal studies. |
| Certified Reference Materials | Commercially available biospecimens with known biomarker values, used to calibrate assays between labs and platforms. |
| Digital PCR Master Mix | Provides absolute quantification without a standard curve, essential for establishing reproducible cutoff values for a clinical assay. |
| Cell Line-Derived Xenograft (CDX) RNA Pool | A consistent, homogeneous biological control for daily run quality control of complex assays like gene expression signatures. |
| Formalin-Fixed, Paraffin-Embedded (FFPE) Control Tissue | Validates assay performance on degraded RNA, which is typical in retrospective clinical cohorts and routine pathology. |
Critical Pathway Visualization
Title: Critical Path for Translational Model Development
Title: Anti-Overfitting Data Partition & Validation Workflow
Conclusion
Effectively addressing overfitting is not a single step but a pervasive mindset that must be integrated into every stage of method development and evaluation. By understanding its foundational causes, employing proactive methodological defenses during model building, diligently troubleshooting performance, and adhering to rigorous validation frameworks, researchers can significantly enhance the robustness and reproducibility of their work. The future of credible biomedical research hinges on this disciplined approach. Moving forward, the adoption of preregistration, shared code and data, and the development of more sophisticated algorithms that inherently penalize complexity will be crucial. Ultimately, defeating overfitting is essential for generating findings that are not just statistically significant but are scientifically meaningful and reliably translatable to improve patient outcomes in drug development and clinical practice.